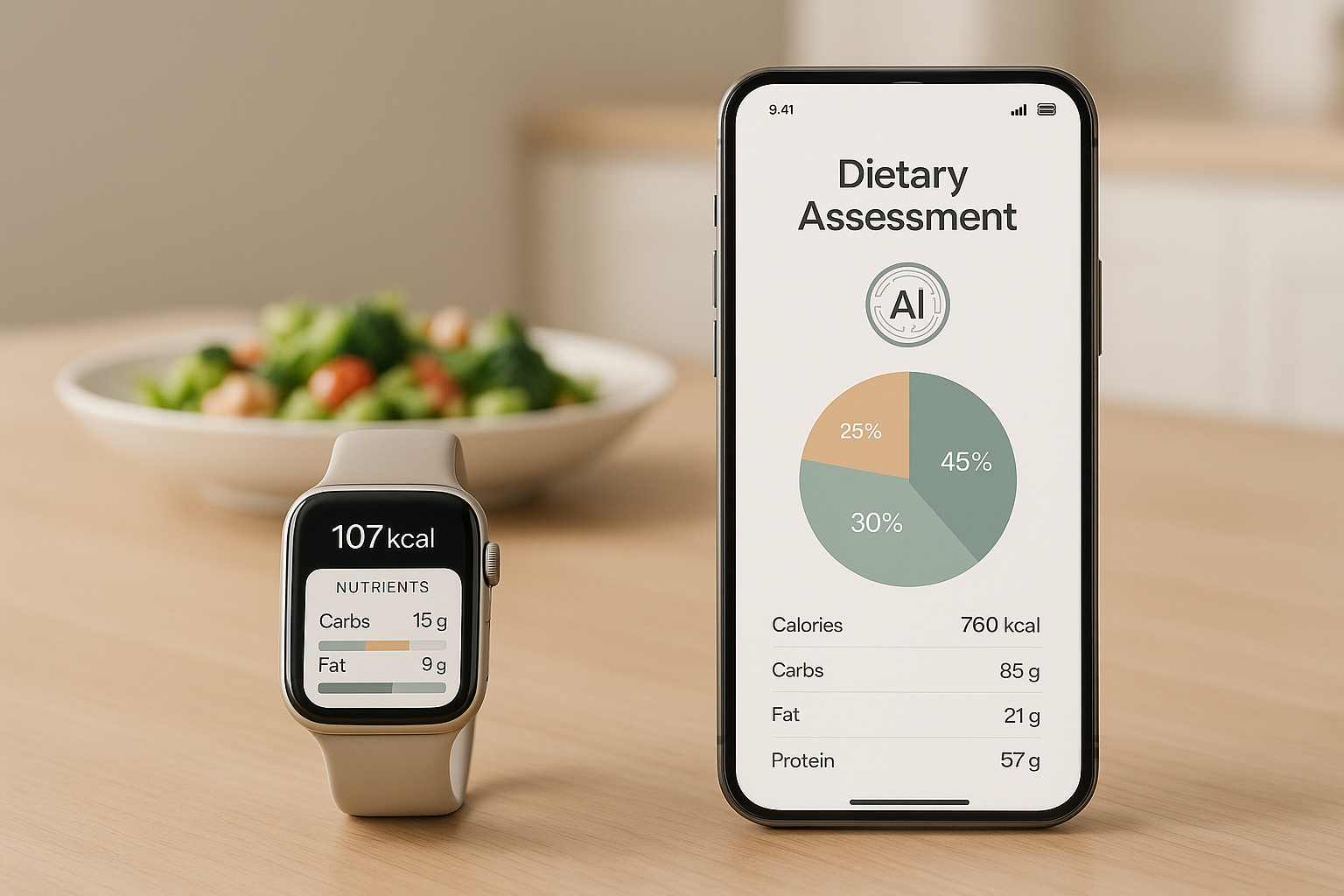
Tracking your food intake is hard. Most methods, like food diaries or questionnaires, rely on memory, which often leads to errors like forgetting snacks or underestimating portions. This makes it tough for dietitians and researchers to get accurate data. AI is changing this by using tools like photo recognition, voice logging, and wearable sensors to make food tracking faster and more precise.
Key takeaways:
- AI tools like image-based apps and sensors reduce errors in food tracking.
- Studies show AI can match or exceed traditional methods in calorie and nutrient estimation, with correlation coefficients often above 0.7.
- These tools save time for clinicians and help patients manage conditions like diabetes or heart disease by offering real-time feedback.
- Platforms like BondMCP combine AI dietary tracking with health data (e.g., sleep, glucose) for personalized insights.
While AI tools are improving accuracy and efficiency, challenges like food database gaps, user fatigue, and validation across diverse populations remain. Future efforts should focus on addressing these issues and integrating dietary tools into broader health systems.
LSS Seminar - Harnessing Artificial Intelligence for Precision Dietary Assessment
Recent Advances in AI-Based Dietary Assessments
In the past few years, dietary assessments have undergone a transformation, shifting from traditional memory-based logs to advanced AI-powered systems. These systems leverage tools like smartphone cameras, wearable sensors, and conversational agents to make tracking food intake more accurate and less burdensome. A systematic review conducted in 2025 revealed that nearly half (46.2%) of AI-based dietary assessment studies now employ deep learning techniques, while 15.3% still use traditional machine learning methods [4]. This shift highlights the growing reliance on neural networks capable of identifying foods and estimating portion sizes with minimal human effort.
These advancements are not confined to theoretical research. The same review found that 61.5% of studies were conducted in preclinical environments, where researchers rigorously test these tools under controlled conditions before introducing them to clinics or consumers. The ultimate aim is to develop systems that integrate seamlessly into daily life - whether you're eating at home, grabbing fast food, or dining at a restaurant. These developments are paving the way for a new generation of AI-driven dietary tools, each designed to address specific challenges in traditional dietary tracking.
Types of AI-Driven Dietary Tools
AI dietary tools have been categorized into several distinct types, each addressing the limitations of older methods like food diaries and 24-hour recalls [6][2][3].
Image-assisted dietary assessment apps are perhaps the most recognizable. These apps allow users to take a photo of their meal, which is then processed by deep learning models - often convolutional neural networks (CNNs) - to identify the foods and estimate portion sizes. This eliminates the need for manual searches in food databases or guessing serving sizes, saving time and reducing errors. Studies indicate that modern CNNs outperform earlier methods in food recognition [2][4].
Wearable sensor systems offer a more passive approach. Devices like smartwatches or specialized sensors monitor motion, audio, heart rate, and other physiological signals to infer eating habits. These systems can detect when you’re eating, estimate meal durations, and even track energy intake. By analyzing these signals, machine learning models identify patterns such as frequent snacking or skipped meals. Some systems pair wearables with mobile apps, prompting users to confirm meals with a quick photo or voice note when eating is detected [6][3].
Conversational agents and AI dietitians use natural language processing to simplify dietary logging. Instead of filling out forms, users can describe their meals in plain language, such as “I had scrambled eggs with toast and coffee.” The AI then identifies the foods, estimates portion sizes, and calculates nutritional content. Advanced systems, often referred to as "AI dietitians", combine language models with image analysis to offer personalized meal plans, interpret dietary data, and provide real-time feedback. These tools have shown promise in managing chronic conditions like type 2 diabetes, where they improve adherence to dietary recommendations and enhance self-monitoring accuracy [6][3].
Hybrid and multimodal systems are becoming increasingly common. These platforms integrate data from multiple sources - smartphone cameras, wearables, voice commands, and contextual information like time of day and location - to create a more comprehensive dietary record. For example, platforms like BondMCP – Health Model Context Protocol unify dietary data with other health metrics, offering personalized recommendations based on a broader context [2][3].
| Tool Type | How It Works | Key Advantage |
|---|---|---|
| Image-based apps | Smartphone photos analyzed by CNNs to identify foods and estimate portions [3][4] | Captures meals in real time with minimal manual input |
| Wearable sensors | Tracks motion, audio, or physiological signals to infer eating occasions and intake [6][3] | Passively detects eating, prompting users only when necessary |
| Conversational AI | Uses natural language input and AI coaching to guide logging and provide feedback [6][3] | Simplifies logging; improves adherence and data quality |
| Hybrid systems | Combines images, sensors, voice, and contextual data for comprehensive tracking [2][3] | Provides a robust and complete dietary record |
By capturing dietary intake closer to the moment of consumption, these tools address common issues like under-reporting, portion size errors, and recall bias. Research shows that AI-based methods significantly reduce the underestimation of energy and macronutrient intake when compared to traditional self-reporting techniques, especially in real-world settings [2][3][4]. These categories highlight the growing potential of AI in dietary assessment.
Main Application Areas
Building on these innovations, AI is now being used in a variety of dietary applications [2][3].
General dietary tracking and weight management: Image recognition apps help users track their food intake with precision, supporting weight management and overall health. Many of these tools integrate with wearables, enabling users to see how their dietary choices align with other health metrics - a feature that resonates strongly in the U.S. market.
Personalized nutrition and cardiometabolic health: Some systems combine dietary data with lab results, such as cholesterol levels, glucose readings, and inflammatory markers. This allows for tailored meal plans that adapt to individual health needs. For instance, if a user’s LDL cholesterol is high, the system might recommend increasing fiber intake and reducing saturated fat, while tracking adherence and updating recommendations based on new lab results [6][3].
Chronic disease management: AI tools are particularly valuable for managing conditions like obesity, type 2 diabetes, and cardiovascular disease. They enable real-time monitoring of food intake, allowing clinicians to detect problematic patterns and intervene more quickly than traditional quarterly appointments. AI dietitians for diabetes, for example, have shown improvements in dietary adherence and physical activity during preclinical evaluations. Some of these systems are now being tested in randomized controlled trials to assess their impact on clinical outcomes like HbA1c levels [2][3].
A 2025 narrative review highlighted that AI systems improve the efficiency and accuracy of nutrition care, particularly when they integrate sensors, wearables, and multimodal data. Controlled studies have reported moderate-to-strong accuracy for image-based assessments and AI coaching [2]. However, the review also noted a lack of extensive data on their effectiveness and user adherence in real-world clinical or community settings, emphasizing the need for larger, more diverse trials.
For U.S. clinicians managing large patient loads in obesity and cardiometabolic clinics, these tools save time by automating data entry and food coding, allowing for more meaningful conversations about behavior change. For researchers, they enable large-scale studies without the labor-intensive process of manually analyzing food diaries. And for individuals managing chronic conditions, AI makes dietary tracking more consistent and accurate [3][7].
As these tools evolve, the next challenge lies in integrating them across platforms. While many systems already combine mobile apps, wearables, lab data, and clinical records to provide personalized dietary recommendations, these components often operate in isolation. A context-orchestration system like BondMCP – Health Model Context Protocol could unify dietary data with sleep, activity, glucose levels, and medication information, ensuring that feedback and recommendations adapt automatically without requiring manual input across multiple tools [5][6][3].
Validation Methods and Study Results
To determine the reliability of AI dietary tools, researchers compare them with established traditional methods. These conventional approaches - such as 24-hour dietary recalls, food frequency questionnaires (FFQs), and weighed food records - serve as benchmarks for validation. The aim is to ensure AI tools deliver results that closely match these trusted methods while requiring less effort.
In validation studies, participants often log their food intake using both an AI-based tool and a traditional method. Researchers then analyze the data from both sources to assess alignment. For instance, a participant might use an image-based app to record meals while also completing a detailed 24-hour recall with a dietitian. By comparing the two datasets, researchers can gauge the AI tool's accuracy. The closer the results, the more confidence researchers have in the tool's performance.
Comparing AI Tools with Standard Methods
Traditional methods are known for their accuracy but can be time-intensive. A 24-hour dietary recall involves participants recounting their food intake through structured interviews with a dietitian, which can yield reliable results but depends heavily on memory. FFQs, on the other hand, ask participants to estimate how often they consume specific foods over a period, making them useful for identifying dietary patterns but less precise for nutrient quantification. Weighed food records, often regarded as the "gold standard", require participants to weigh and log every item they consume. While highly accurate, this method is impractical for everyday use outside research settings.
AI dietary tools aim to replicate the precision of these methods while minimizing the workload for users and clinicians. Instead of relying on memory or manually weighing food, AI tools utilize technologies like smartphone cameras, wearable sensors, or conversational interfaces to capture real-time data. Studies suggest that in free-living conditions, AI tools often match or even surpass traditional recalls in accuracy, thanks to automated data coding and nutrient analysis.
However, these tools perform best in controlled environments where foods are easily identifiable and well-represented in their training datasets. In real-world scenarios - particularly with mixed dishes, varied cuisines, or foods uncommon in U.S. databases - their accuracy can decline.
| Aspect | Standard 24-hour Recall / FFQ | Weighed Food Record | AI-Based Dietary Tools |
|---|---|---|---|
| Reference standard use | Commonly used for validation [6][2] | Gold standard for accuracy [8][2] | Validated against recalls or weighed records [6][8] |
| Participant burden | Moderate; memory-dependent [2] | High; requires weighing all food [8][2] | Low; uses photos or quick mobile entries [8][4] |
| Clinician workload | High; manual data processing [2] | Very high; labor-intensive [8][2] | Lower; automated data analysis [6][2] |
The table highlights how AI tools reduce the workload for both participants and clinicians, a crucial advantage in U.S. healthcare settings where dietitians often manage large patient loads. Automation allows clinicians to focus on interpreting results and guiding patients toward healthier habits.
Key Validation Metrics
Researchers rely on specific metrics to measure AI tool accuracy. The correlation coefficient, for example, evaluates how closely AI-generated estimates align with traditional methods. For energy and macronutrient intake, correlation coefficients typically fall between 0.6 and 0.9, indicating moderate to strong agreement [6]. Bland-Altman analysis further examines this agreement, identifying whether AI tools systematically overestimate or underestimate intake.
Another metric, mean absolute percentage error, calculates the average difference between AI estimates and reference values. For instance, a study using a convolutional neural network for image-assisted dietary assessment reported a relative error of about 31.2% (roughly 90 kcal) for calorie estimates and 36.1% (approximately 0.7 g) for salt compared to weighed food records [8]. AI tools generally estimate portion sizes with errors ranging from 20–40%, with clearer foods yielding more accurate results than mixed or amorphous items [6][8].
Macronutrient estimates (protein, carbohydrates, fat) tend to perform well, with correlation coefficients often between 0.6 and 0.8 [6][8]. However, micronutrient estimation is less reliable due to the need for highly detailed food composition data.
In clinical or real-world settings, AI-powered tools have demonstrated "fairly accurate" results, often within 10–20% of reference methods, for estimating energy and macronutrient intake in patients managing conditions like obesity, diabetes, or dementia [2][4]. This level of accuracy is generally sufficient for identifying dietary issues and guiding interventions, while significantly reducing the time clinicians spend on data collection.
That said, challenges remain. AI tools often perform less accurately in uncontrolled, everyday conditions where factors like lighting, camera angles, and food presentation can interfere. Moreover, many validation studies focus on high-income countries with relatively uniform food habits, limiting their relevance to diverse U.S. populations.
For robust validation, researchers emphasize the need for strong reference methods - such as multiple-day weighed food records or repeated 24-hour recalls - and tools like Bland-Altman plots. Additionally, AI tools should use up-to-date U.S. food composition databases, like USDA FoodData Central, and clearly outline how food data is mapped to nutrient profiles [6].
As these tools advance, future validation efforts will need to go beyond accuracy metrics. Researchers must also assess their reliability in real-world settings, user adherence, and clinical impact. Randomized controlled trials are beginning to explore whether AI dietary tools can improve outcomes like weight loss or glycemic control.
When integrated with broader health platforms like BondMCP, validation becomes even more complex. These tools must not only provide accurate dietary assessments but also synthesize data from wearables, lab results, and other health metrics. Systems like BondMCP - Health Model Context Protocol ensure that dietary data is contextualized alongside factors like sleep, physical activity, glucose levels, and medications, offering a more comprehensive view of health.
AI-Driven Improvements in Accuracy and Efficiency
AI-powered dietary tools are transforming how we assess food intake and streamline clinical workflows. Advances in computer vision, machine learning, and real-time data processing are addressing challenges that have long plagued traditional dietary assessments. These developments are paving the way for more precise portion measurements and better recognition of dietary patterns.
Better Portion-Size Estimation and Pattern Recognition
Estimating portion sizes has always been tricky in both nutrition research and clinical settings. When people rely on memory or vague measures like "a handful" or "a cup", inaccuracies creep in quickly. AI steps in by using plate segmentation, 3D volume estimation, and shape recognition to analyze photos, distinguish food groups, and calculate portion sizes in grams and calories.
Studies show that AI-based methods are effective at estimating energy and macronutrient intake, making them a practical alternative to traditional approaches. For instance, an AI app designed to evaluate a 2:1:1 plate model (two parts vegetables, one part protein, one part carbohydrates) outperformed both nutrition students and registered dietitians in accurately assessing proportions [9]. By focusing on precise measurements of areas and volumes, these tools help eliminate the variability often seen in human judgment.
Using deep learning and computer vision, image-based AI systems can identify foods from photos, significantly reducing the recall bias typical of self-reported dietary data [2][3][4]. While micronutrient estimation still poses challenges, these tools excel at accurately measuring energy and macronutrient intake [4].
AI doesn't stop at single meals. Machine learning algorithms can analyze timestamped logs and wearable data to detect patterns like late-night snacking or irregular protein consumption. These insights enable personalized, real-time dietary adjustments. By clustering users into dietary profiles linked to metabolic markers, weight trends, or chronic disease risks, these tools provide a more comprehensive view than a single 24-hour recall ever could. For example, integrating data from continuous glucose monitors or sleep trackers with eating patterns can help refine nutritional recommendations. These detailed insights not only address common errors in dietary reporting but also guide clinical interventions and tailored feedback.
Clinical Applications and Efficiency Gains
The precision offered by AI in dietary assessments translates directly into clinical efficiency. Automated dashboards now summarize nutrient trends and adherence scores, saving clinicians valuable time [2][3]. Diabetes and weight-management clinics, in particular, have embraced AI-powered food logging apps, where patients upload meal photos via smartphones. Dietitians can then review weekly summaries, meal timing patterns, and incomplete logs during follow-ups, reducing time spent on charting and allowing for more focused consultations.
In cardiometabolic risk clinics, AI tools integrated with electronic health records (EHRs) automatically flag dietary risk factors, such as frequent high-sodium or high-saturated-fat meals. This allows clinicians to quickly verify and discuss these issues [1][2][3]. These systems also support remote monitoring and telehealth appointments, which are especially beneficial for patients in rural areas or those with limited mobility. A review highlighted that AI-assisted dietary tools can estimate energy and macronutrient intake in individuals with conditions like obesity, diabetes, and dementia while being "non-laborious, time-efficient, [and] user-friendly" [3]. Features like gamification, push notifications, and conversational agents further improve user engagement, leading to more consistent logging and better data [2][3][7].
AI-powered virtual dietitians and coaching systems have shown promise in improving adherence to dietary recommendations for conditions like obesity, type 2 diabetes, and cardiometabolic risk [2][3][7]. Automated feedback - such as reminders to log meals, tips for balancing macronutrients, or alerts for excessive sodium intake - helps patients stay on track between appointments, easing the workload for both patients and clinicians.
These tools work best when trained on high-quality food image datasets and nutrient databases that reflect common U.S. foods, regional cuisines, branded products, and restaurant meals [2][3][4]. Regular updates and simple tutorials - teaching users how to take clear, well-lit food photos with reference objects - help reduce errors. Additionally, ensuring these tools are validated across diverse demographic groups is essential to ensure they work effectively for all users [2][3][4].
When integrated with platforms like BondMCP – Health Model Context Protocol, these AI tools transform dietary data into actionable health interventions. For example, a pattern of late-night high-carb meals could prompt adjustments to a patient's coaching plan or supplement regimen - or trigger a reevaluation of glycemic targets. This integration ensures that dietary data contributes to real-time care decisions rather than sitting idle in a patient portal. For U.S. clinics and digital health developers, using structured protocols and health-specific ontologies can eliminate fragmented experiences, reduce duplicate assessments, and create a unified system that connects dietary data with other health metrics like sleep, activity, lab results, and medications.
As these tools continue to evolve, they are becoming even more integrated into broader health strategies. Current evidence confirms that AI-based dietary assessments are reliable and effective for estimating energy and macronutrient intake [2][4]. As they mature, these tools are poised to make personalized nutrition more precise and accessible for patients and providers across the U.S. health system.
sbb-itb-f5765c6
Integrating AI Dietary Tools with Personalized Health Platforms
Building on the accuracy of dietary assessments, integrating AI-powered dietary tools into broader health platforms takes personalized nutrition to the next level. By connecting food logs, nutrient tracking, and eating patterns with electronic medical records and remote monitoring systems, these tools can deliver tailored recommendations based on real-time biometrics. This integration creates opportunities for more precise clinical interventions and individualized nutrition strategies.
AI in Chronic Disease and Personalized Nutrition
For those managing conditions like obesity, type 2 diabetes, and cardiovascular disease, AI-driven dietary tools are reshaping how care is delivered. By combining dietary data with metrics like continuous glucose monitoring, lipid profiles, blood pressure readings, and activity levels from wearables, clinicians can create nutrition plans that evolve alongside a patient’s changing health needs.
Consider this: a randomized controlled trial using an AI-enhanced nutrition app showed participants increased their fruit and vegetable intake while cutting back on sugary drinks over three months compared to a control group [6]. For diabetes and cardiometabolic diseases, AI systems can analyze logged meals alongside post-meal glucose or lipid responses. This helps identify problematic foods, suggest healthier alternatives, and generate summaries for clinicians to adjust medications or provide targeted dietary advice [6][3].
In weight management, AI tools track calorie intake and energy density, adapting goals and offering automated coaching as weight and activity data shift [6][3]. To integrate these tools into U.S. healthcare, patients must enroll in an AI-enabled app, grant data-sharing permissions, and link the app to their clinic’s platform for dietitian review during visits. Clinical teams can set specific alert thresholds - like high sodium or added sugar levels - and translate AI insights into actionable plans [6][3].
However, challenges persist. User-entered logs can vary in quality, and many tools lack validation across diverse cuisines and populations. Additionally, there are issues with interoperability between consumer apps, clinical systems, and insurance platforms. Addressing these gaps requires using tools with validated data, standardizing formats, offering clear logging instructions, and ensuring oversight by dietitians or clinicians [6][4].
BondMCP as a Context-Orchestration Layer
A unified context layer, like BondMCP, helps solve the problem of fragmented health data. Often, food logs, sleep tracking, lab results, and supplement protocols exist in isolation, making it difficult to provide cohesive care. BondMCP addresses this by creating a shared health context, allowing dietary data from AI tools to integrate seamlessly with wearables, lab biomarkers, and other health metrics [5].
BondMCP brings diverse health data into a coordinated system. For instance, when an AI dietary tool sends meal logs and nutrient estimates to BondMCP, the platform can integrate this information with lipid profiles, triggering updates to cardiovascular risk assessments and dietary recommendations. It can even refine supplement suggestions while syncing with exercise and sleep data [5].
Imagine someone with type 2 diabetes using an AI food-logging app and a continuous glucose monitor. BondMCP can detect patterns, such as high post-meal glucose linked to specific foods, and coordinate actions like suggesting healthier alternatives or notifying the clinical team if thresholds are repeatedly exceeded [5][6]. For individuals focused on longevity, BondMCP can combine dietary quality scores, body composition trends, inflammatory markers, and sleep data to adjust protein targets, meal timing, or supplement plans - all in pursuit of long-term health goals.
BondMCP’s technical framework, including its structured protocol and SDK, allows developers to build interoperable and health-aware tools without starting from scratch. For U.S. clinics and digital health developers, using standardized data formats ensures that dietary assessments can integrate smoothly with other health metrics like sleep, activity, lab results, and medications.
Privacy and governance are critical considerations. Given that dietary logs, biometrics, and lab data contain sensitive information, clear patient consent, transparent data-use policies, and strong security measures are essential to comply with U.S. regulations. Ethical practices are equally important - teams must monitor for algorithmic bias, avoid over-reliance on automated suggestions, and ensure recommendations are explainable and backed by clinical oversight [5][6].
Success in integrating AI dietary tools will ultimately be measured by clinical outcomes - such as improved HbA1c levels, blood pressure, lipid profiles, or weight - and operational efficiency, like reduced time spent on manual dietary assessments and better patient adherence. By connecting multiple data streams through a context-orchestration layer, platforms like BondMCP highlight the move toward integrated health systems that link diet with other health factors. This approach supports both chronic disease management and the pursuit of long-term wellness [5][6][3].
Limitations and Future Directions
AI-powered dietary assessment tools hold great promise, but several challenges must be addressed before they can deliver accurate and meaningful results for all U.S. populations. Whether you're a patient, clinician, or developer, understanding these limitations is crucial.
Current Limitations
There are several barriers that restrict the effectiveness of current AI dietary tools:
- Image quality and technical requirements: These tools often rely on smartphone photos to identify foods and estimate portions. Poor lighting, mixed dishes like casseroles, or awkward camera angles can significantly reduce their accuracy. This dependency means the tools work best for users with newer smartphones, stable internet connections, and familiarity with technology [2][4].
- Limited and non-diverse food databases: Many systems are trained on images and dietary habits from higher-income regions like North America, Europe, or East Asia. This creates a gap in recognizing foods from underrepresented groups, such as Black, Hispanic/Latino, Native American, and rural communities. For example, dishes like Southern soul food or Mexican-American home cooking may be misclassified due to a lack of representation in training datasets [2][3].
- Micronutrient tracking challenges: While these tools can reasonably estimate macronutrients like calories and protein, they struggle with micronutrients such as vitamins and minerals. This is especially problematic for individuals managing conditions like anemia or osteoporosis, where precise micronutrient data is critical [2][4].
- User fatigue: Requiring users to photograph every meal, correct errors, and review feedback often leads to frustration and eventual dropout. Clinicians face similar issues, as these tools often lack integration with electronic health records and produce reports that are time-consuming to interpret [2][3].
- Privacy and bias concerns: Image-based tools may inadvertently capture sensitive information, such as household environments or personal details. Concerns about how this data is shared or used - by insurers, employers, or advertisers - can deter users. Additionally, algorithmic bias can lead to better performance for certain cuisines or body types, raising fairness issues [5][2].
- Lack of standardized validation: Many studies validating these tools are inconsistent and prone to bias, making it difficult to generalize their accuracy across diverse U.S. populations. Without rigorous, independent testing in real-world settings, performance claims remain limited to small, homogenous samples [2][4].
Opportunities for Future Research
Addressing these challenges requires focused research and development:
- Diversifying training datasets: Expanding datasets to include underrepresented racial, ethnic, and socioeconomic groups is critical. Future studies must actively recruit participants from these communities to ensure AI tools work effectively for all Americans, not just those with access to the latest technology [2][3].
- Building U.S.-specific food databases: Curating labeled datasets of foods from various traditions - such as Southern, Caribbean, and Native American cuisines - can improve recognition. Collaborating with community groups, local restaurants, and public health programs will be key. Advanced techniques like active learning could allow tools to improve accuracy through user corrections while maintaining privacy [2][3].
- Using multimodal inputs: Relying solely on photos limits accuracy. Combining image recognition with barcode scanning, voice input, and wearable sensor data can provide a fuller picture. For example, when a photo isn't clear, the app could prompt users to confirm ingredients or portion sizes through quick questions, enhancing nutrient estimates [2][3].
- Linking to clinical outcomes: More research is needed to connect AI-derived dietary metrics to health outcomes like weight, blood sugar levels, and blood pressure. Large, multi-center trials in diverse U.S. populations could compare AI-supported dietary tools with standard care over several months to evaluate their impact on chronic conditions [2][4].
- Improving integration with health systems: Combining dietary data with electronic health records, lab results, and wearable metrics could enable tailored nutrition plans for conditions like diabetes and cardiovascular disease. For instance, a system could use food logs, glucose monitor data, and lipid panels to suggest meals and generate actionable summaries for clinicians [5][2].
- Focusing on transparency: Both patients and clinicians need clear explanations for AI-generated recommendations. Better documentation of training data, subgroup performance, and compliance with privacy regulations can build trust and encourage adoption [5][2].
- Testing in real-world settings: Deploying these tools in primary care clinics, community health centers, and workplace wellness programs can reveal how they perform at scale. Pragmatic trials can also explore whether they reduce clinician workload and improve patient outcomes [2][3].
Conclusion
AI has reshaped how we approach dietary assessment. Traditional methods, which relied heavily on memory and manual tracking, often fell short due to under-reporting and inaccuracies. In contrast, AI-based tools now automate food recognition using smartphone images, provide real-time tracking, and minimize recall bias. This makes it easier to align dietary habits with metabolic data, lab results, and personal health goals.
Studies show that AI tools are highly accurate, with correlation coefficients often exceeding 0.7 - and reaching as high as 0.97 in controlled environments [4]. This level of precision not only boosts adherence but also supports real-time dietary adjustments, which are especially critical for managing conditions like obesity and diabetes. The detailed, long-term data these tools provide allow healthcare professionals to fine-tune medications and dietary plans, advancing preventive and value-based care across the U.S.
The real power of AI dietary tools emerges when they are part of a larger, interconnected health system. For example, BondMCP integrates dietary data with other health metrics like continuous glucose monitoring, lab results, activity tracking, and sleep patterns. This means a U.S. user can track how a specific breakfast influences their blood sugar, energy levels, and sleep quality - enabling automated, personalized dietary recommendations without the need for manual data entry.
To make these tools even more effective, systems must support secure API integrations, standardized food data (including units and timestamps), and HIPAA-compliant data handling. Simplifying the user experience - through features like photo-based logging and passive data collection - and presenting insights in familiar U.S. measurement units with culturally relevant meal examples can further enhance usability.
That said, challenges remain. AI dietary tools still struggle with accurately capturing micronutrient intake and may reflect biases from limited training datasets. Expanding validation studies to include diverse, free-living U.S. populations is essential. Pairing these tools with clinician oversight, user feedback, and context-aware platforms like BondMCP can help address these limitations and improve their performance over time.
Looking ahead, AI systems will continue to evolve. Advances in food recognition, combined with genomic and physiological data integration, will create even more personalized and clinically validated platforms. These improvements could lead to broader reimbursement, inclusion in care guidelines, and a stronger role in precision nutrition strategies.
FAQs
How does AI improve food recognition and portion estimation for diverse diets?
AI is transforming how dietary assessments are conducted, leveraging image recognition and machine learning to identify foods and estimate portion sizes with impressive precision. These systems are trained on vast datasets that cover a broad range of cuisines, enabling them to recognize countless dishes and ingredients.
By analyzing meal photos, AI can pinpoint food types, measure portion sizes, and even break down the nutritional content. This detailed analysis allows for tailored dietary recommendations, simplifying the path for individuals looking to improve their health and reach specific wellness objectives.
What challenges do AI tools face in accurately tracking micronutrients in diets?
AI dietary tools encounter several hurdles when it comes to tracking micronutrients with precision. A key challenge lies in the variability of food composition data. Nutrient levels can fluctuate significantly depending on factors like the food's source, how it's prepared, and the portion size. This inconsistency makes it tricky for AI systems to deliver accurate nutrient estimates tailored to each individual.
Another obstacle is the dependence on self-reported data, such as food diaries or meal logs. These are often riddled with inaccuracies - people might forget to log certain foods, misjudge portion sizes, or leave out critical details. These gaps can throw off the micronutrient calculations, leading to less reliable results.
To tackle these issues, AI systems are turning to cutting-edge tools like image recognition, natural language processing, and wearable device integration. These advancements aim to provide deeper insights into dietary habits and offer more personalized recommendations.
How can AI tools improve and support personalized nutrition in healthcare systems?
AI-powered tools are transforming personalized nutrition by analyzing dietary habits, spotting nutritional deficiencies, and customizing recommendations to fit individual health needs. These tools work smoothly with healthcare platforms, giving providers the insights they need to design effective nutrition plans.
With AI, healthcare systems can pull data from multiple sources - such as fitness trackers, lab reports, and food diaries - to provide precise, real-time advice. This approach not only boosts efficiency but also ensures that nutrition strategies align with a person’s specific health goals and conditions.