AI and epigenetic clocks are reshaping healthcare by focusing on biological age, a reflection of how well your body is functioning, rather than just your chronological age. These clocks analyze DNA methylation patterns to predict aging and health risks, offering insights far beyond traditional health metrics. Here's what you need to know:
-
Types of Epigenetic Clocks:
- Horvath Clock: Estimates chronological age using 353 CpG sites.
- PhenoAge: Predicts functional aging and health risks using clinical biomarkers.
- GrimAge: Assesses cumulative damage and mortality risk, excelling at predicting cardiovascular and cancer risks.
- DunedinPACE: Tracks the rate of aging, useful for short-term changes from lifestyle or treatments.
-
AI's Role:
- Machine learning refines predictions by analyzing vast DNA data, uncovering patterns traditional methods miss.
- Tools like MAPLE achieve high accuracy (median error of 1.6 years) and integrate data from wearables, lab tests, and health records.
- AI creates "EpiScores" to predict health risks like inflammation or insulin resistance before symptoms appear.
-
Preventive Health Benefits:
- Early detection of health risks, such as prediabetes or cardiovascular issues.
- Personalized interventions based on biological age and lifestyle tracking.
- Real-time feedback on how habits like sleep and diet impact aging.
While challenges like technical variability and population bias remain, these tools are paving the way for more personalized, proactive healthcare. Biological age is becoming a key metric for optimizing health and longevity.
Steve Horvath on Epigenetics, Aging, and Reversing the Biological Clock | WIRED Health
sbb-itb-f5765c6
What Are Epigenetic Clocks and Their Main Types
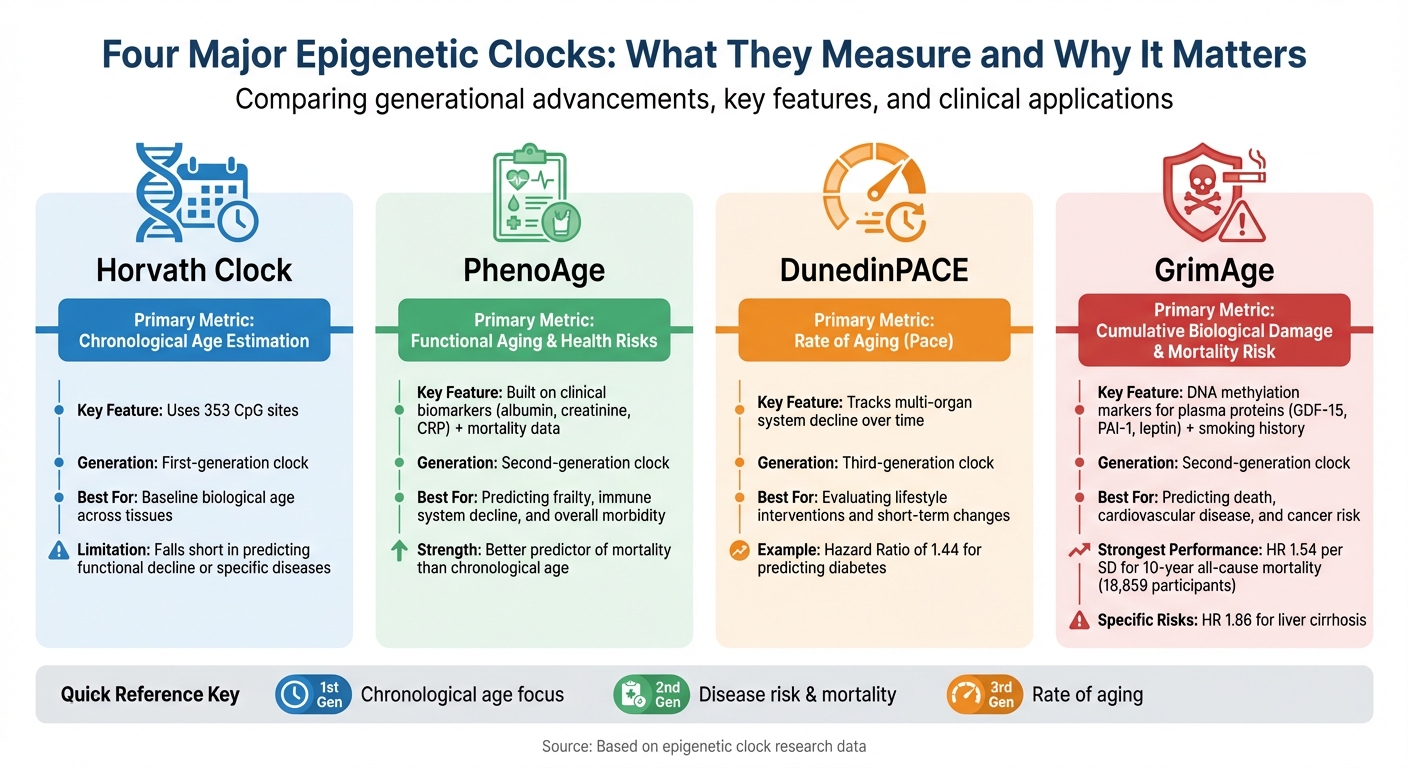
Comparison of Four Major Epigenetic Clock Types and Their Applications
Epigenetic clocks have progressed over time to tackle different aspects of aging. First-generation clocks, like Horvath, are designed to predict your chronological age with high precision. Second-generation clocks, such as PhenoAge and GrimAge, take it a step further by estimating disease risk and mortality. Third-generation clocks, like DunedinPACE, focus on something entirely new - the rate at which you're aging right now. Below, we’ll explore the standout clocks from each generation.
Horvath Clock and PhenoAge
The Horvath Clock relies on 353 CpG sites to estimate chronological age across various tissues [3]. While it’s highly reliable for determining chronological age, it falls short in predicting functional decline or specific diseases [7][10]. Think of it as a baseline tool - excellent for measuring chronological age but not for assessing health risks.
On the other hand, PhenoAge is built on clinical biomarkers like albumin, creatinine, and CRP, along with mortality data. This clock excels at predicting health issues such as frailty, immune system decline, and overall morbidity [7][9]. As Brandon Warner from Verseon International Corporation explains:
"Phenotypic clocks have shown to be better predictors of mortality than chronological age and do so using easily measurable clinical variables."
PhenoAge is particularly effective for evaluating functional aging and metabolic stress, making it a practical choice for understanding long-term health [9].
DunedinPACE and Aging Rate Measurement
While PhenoAge focuses on functional aging through clinical markers, newer models like DunedinPACE measure the dynamic pace of aging - essentially, how quickly your body is wearing down over time [3][7]. Unlike most clocks, which provide a static biological age, DunedinPACE tracks the rate of multi-organ system decline. This makes it especially useful for monitoring short-term changes due to lifestyle adjustments or medical treatments. For example, in a large study, DunedinPACE showed a Hazard Ratio of 1.44 for predicting diabetes [10]. It’s an excellent tool for determining whether your diet, exercise, or treatments are slowing down the biological aging process. This is a core focus of personalized longevity AI agents that translate these metrics into actionable advice.
GrimAge and Cumulative Damage Assessment
GrimAge stands out as one of the most reliable predictors of mortality among epigenetic clocks. It incorporates DNA methylation markers for plasma proteins like GDF-15, PAI-1, and leptin, along with smoking history [7][10]. In the Generation Scotland study, which included 18,859 participants, GrimAge v2 demonstrated the strongest link to 10-year all-cause mortality, with a Hazard Ratio of 1.54 per standard deviation of age acceleration [10]. It’s particularly adept at predicting risks for cardiovascular disease, cancer, and liver conditions like cirrhosis (HR 1.86) [10]. By reflecting cumulative biological damage, GrimAge provides a robust assessment of long-term health risks [7].
| Clock Type | Indicator | Application |
|---|---|---|
| Horvath | Chronological age estimation | Baseline biological age across tissues [3] |
| PhenoAge | Clinical biomarkers & mortality | Predicting frailty and immune decline [7] |
| DunedinPACE | Rate of aging (pace) | Evaluating lifestyle interventions [7] |
| GrimAge | Cumulative biological damage and mortality risk | Predicting death, CVD, and cancer risk [7][10] |
These diverse epigenetic clocks form the foundation for exploring how AI can refine these models to deliver more personalized and preventive healthcare solutions.
How AI Improves Epigenetic Clocks
Linear models often fall short in capturing the complex relationship between DNA methylation and aging. Machine learning, on the other hand, uncovers hidden patterns and non-linear interactions across thousands of CpG sites. As Joshua J. Levy explains:
"Machine learning technologies offer promising avenues to enhance our understanding of the biological mechanisms governing aging by further elucidating the gap between biological and chronological ages." [2]
AI for Data Analysis and Pattern Detection
AI thrives in handling the vast datasets generated by DNA methylation arrays, which can measure over 900,000 CpG sites [2]. Techniques like CART and deep learning reveal non-linear associations that traditional models miss [1] [2]. For example, the MAPLE framework employs Siamese networks to process DNA methylation data and adjust for batch effects [6]. MAPLE has proven its accuracy, achieving a median absolute error of 1.6 years across 31 benchmark tests and demonstrating strong predictive capabilities, with an AUROC of 0.98 for stroke risk and 0.85 for pre-disease detection [6].
Another standout is EnsembleAge, which combines multiple penalized regression models to reduce bias. This method achieved a robust correlation with chronological age (R = 0.98) [4]. By integrating various models, these systems provide more reliable biological age estimates than single-model approaches.
AI also enhances transparency. Tools like Shapley Additive Explanations (SHAP) help researchers identify which CpG sites or biological pathways - such as PRC2 target sites - contribute to age acceleration [1] [2] [3]. This level of detail is crucial for understanding why someone's biological age might differ from their chronological age and determining actionable interventions. These insights are essential for developing patient-centered treatment plans tailored to individual needs.
These advanced tools pave the way for combining diverse health data streams.
Combining AI with Wearables and Omics Data
AI doesn't stop at DNA methylation. It now integrates data from wearable devices, medical imaging, and other "omics" fields like transcriptomics, proteomics, and metabolomics to refine biological age estimates [2] [3]. This multimodal approach allows for real-time health insights and tracking of biological age, showing users how everyday habits - like sleep, exercise, and diet - affect their aging process [3]. In clinical settings, biological age could serve as a "next-gen vital sign", complementing traditional metrics like blood pressure and BMI [3].
AI can also generate "EpiScores", synthetic indicators derived from DNA methylation patterns, to predict protein levels and clinical markers like IL-6 or HbA1c. These insights help assess specific health risks more effectively [3].
Case Study: CXR-Age from Medical Imaging
AI's reach extends beyond DNA methylation. For example, imaging-based models like CXR-Age use chest X-rays to identify aging biomarkers. While DNA-based clocks remain the gold standard, imaging provides a broader view by assessing organ-level decline [13] [2]. Since medical imaging is often more accessible than DNA sequencing, it offers a practical tool for large-scale biological age screening [2].
As Joshua J. Levy highlights:
"DNAm-derived age acceleration can be predicted from these other data types [imaging] to uncover additional aging features and can serve as lower-cost surrogate assessments." [2]
The EHRFormer model, a transformer-based AI, predicts biological age using imaging and electronic health records. In the UK Biobank cohort, it achieved a Mean Absolute Error of 4.14 [13]. These multimodal frameworks not only improve age precision but also enhance disease risk prediction [13] [7]. By filtering out visible signs of aging, they reveal deeper biological age acceleration that might otherwise go unnoticed [2].
| Feature | Traditional Linear Clocks | AI-Enhanced Clocks |
|---|---|---|
| Data Handling | Limited to linear CpG-age relationships [2] | Captures non-linear interactions and patterns [1] |
| Generalizability | Sensitive to batch effects [6] | Reduces bias with pairwise learning or ensembles [4] [6] |
| Predictive Power | High for chronological age [3] | Excels at predicting age-related diseases [6] [11] |
| Interpretability | Simplified but intuitive [2] | Requires tools like SHAP for deeper insight [1] [2] |
Uses in Preventive Healthcare
AI-powered analytics are transforming preventive healthcare by enabling proactive and personalized interventions. Instead of waiting for symptoms to appear, these tools can identify health risks years - or even decades - before diseases manifest. One key concept is age acceleration, which occurs when biological age surpasses chronological age. This serves as an early warning for potential health deterioration and increased mortality risk [1][2].
Personalized Interventions and Early Risk Detection
Advanced frameworks like MAPLE are making it possible to detect "pre-disease" states before clinical symptoms arise. For instance, MAPLE has successfully identified early markers for conditions such as Systemic Insulin Resistance, Prediabetes, and cardiovascular issues like Coronary Artery Ectasia, all before significant health events occur [6]. A striking example comes from the LinAge2 clinical clock, which flagged a 72-year-old man as biologically 16 years older than his actual age, predicting his death from diabetes 5.4 years later [11]. As highlighted by NPJ Aging:
"Aging clocks should similarly enable early detection of hidden or subclinical diseases, surpassing the capabilities of diagnostics by identifying disease processes years or decades before overt disease is present." - NPJ Aging [11]
AI also generates EpiScores, synthetic indicators derived from DNA methylation patterns. These scores predict protein concentrations and clinical markers like IL-6 or HbA1c, offering insights into risks such as chronic inflammation or insulin resistance long before traditional tests can detect them [3].
Explainable AI for Clinical Decision-Making
For doctors to trust AI models, transparency is essential. Tools like SHAP (Shapley Additive Explanations) provide clarity by pinpointing which specific CpG sites or biological pathways - such as p53 signaling or DNA damage response - contribute to a patient's risk profile. This turns abstract data about biological age into actionable insights for healthcare providers [6][2].
In December 2025, researchers at the Marcus Institute for Aging Research shared findings from a study involving 2,097 adults in the Project Baseline Health Study. They compared a deep-learning model called CXR-Age, which analyzes chest X-rays, with DNA-based clocks like Horvath and PhenoAge. The AI model outperformed DNA clocks in predicting coronary calcium levels and declining lung function, particularly in middle-aged adults [8]. Dr. Douglas P. Kiel, a senior scientist at the institute, noted:
"AI tools like this could become an important complement to traditional risk assessments... revealing how our organs are aging before symptoms develop." [8]
When combined with real-time health data, these tools significantly enhance the scope of preventive care.
Health Optimization with BondMCP
BondMCP takes epigenetic insights and integrates them into everyday health management. By connecting wearables, lab results, and epigenetic measurements, it creates a coordinated system for tracking how daily habits - like sleep quality, exercise, and nutrition - affect your aging trajectory in real time. For example, your sleep tracker might adjust your training routine, lab results could refine your supplement plan, and your epigenetic age could inform longevity strategies - all seamlessly managed by AI.
This system also uses computational models to simulate how specific lifestyle changes might influence future health outcomes [14]. As a result, biological age is emerging as a "next-generation vital sign", complementing traditional metrics like blood pressure and BMI [7][3]. This approach aligns with the World Health Organization's focus on maintaining "Intrinsic Capacity", which emphasizes physical, cognitive, and psychological resilience as key factors in overall healthspan.
Challenges and Future Directions
AI-powered epigenetic clocks still face several obstacles before they can become a part of routine clinical practice. One major issue is technical variability. Differences across measurement platforms and batch effects often introduce noise, which can blur the accuracy of the biological signals these clocks are designed to detect [3][15][6].
Another challenge is population bias, which limits how broadly these models can be applied. Most of these clocks are trained primarily on data from Western populations, making them less reliable for diverse groups [3]. Additionally, many clocks struggle to differentiate between normal aging and disease. For instance, an undiagnosed condition, such as heart failure, might artificially inflate a person’s biological age, reflecting hidden illness rather than actual aging [11]. The LinAge2 research team highlighted this issue:
"Aging clocks should similarly enable early detection of hidden or subclinical diseases, surpassing the capabilities of diagnostics by identifying disease processes years or decades before overt disease is present" [12].
These technical and demographic challenges underline the need for better data integration and standardization to improve reliability and applicability.
Clinical Validation and Model Standardization
One promising way to address these challenges is by integrating multi-omics data. Combining DNA methylation data with information from proteomics, metabolomics, and clinical records can provide a more complete picture of an individual’s health [3][16]. Advanced models like EnsembleAge are already showing how this approach can improve accuracy [4].
Adding Functional Biomarkers to Epigenetic Models
Even with integrative approaches, standardization is critical for clinical use. Consistent preprocessing methods, clear regulatory guidelines, and explainable AI tools (like SHAP) are essential. These measures can help clinicians understand the reasoning behind a model’s predictions, fostering trust and usability in healthcare settings [1][6].
Creating Standard Protocols for AI-Driven Health Systems
Platforms such as BondMCP offer a potential path forward by connecting epigenetic data with other sources, like wearable devices, lab results, and lifestyle information. This kind of integration can transform fragmented data into actionable insights. However, without established protocols and regulatory frameworks, these tools risk being confined to research rather than making a real impact in clinical environments [3].
Conclusion
AI-powered epigenetic clocks are changing the way we think about preventive healthcare. By identifying non-linear aging trends, these tools allow clinicians to detect potential health risks long before symptoms emerge. This approach shifts the focus toward proactive care, driven by integrated data insights.
For example, a 5-year increase in GrimAge acceleration is linked to a 44% higher risk of all-cause mortality and a 33% greater risk of cardiovascular death [5]. On the technical side, AI frameworks like MAPLE have demonstrated remarkable accuracy, with a median absolute error of just 1.6 years across 31 different testing scenarios [6]. This level of precision takes biological age from being an abstract idea to a measurable metric that, as Hidekazu Yamada puts it, "can be incorporated into daily clinical practice as a new 'next-generation vital sign'" [7].
What’s even more transformative is the integration of this precision into a broader health ecosystem. When epigenetic data is combined with wearables, lab results, and lifestyle tracking, it creates a unified view of health. Platforms like BondMCP exemplify this by connecting sleep data, training programs, and lab results into one seamless system. This unified approach replaces scattered apps with AI-driven solutions that optimize health decisions, from adjusting supplement protocols to fine-tuning fitness plans.
Of course, challenges remain. Technical variability and the lack of standardized protocols still hinder widespread clinical use. But as Douglas P. Kiel observed, "AI tools like this could become an important complement to traditional risk assessments" [8]. The potential to transform healthcare is undeniable; the real hurdle lies in speeding up validation and deployment.
Even with these obstacles, the immediate benefits are clear. Biological age feedback not only encourages healthier lifestyle changes but also provides tangible proof of intervention success [3]. The opportunities for patients, clinicians, and researchers are already here.
FAQs
How do I know which epigenetic clock is right for me?
When deciding on an epigenetic clock, it's essential to consider how well it predicts health risks and age-related outcomes, along with its level of clinical validation. Popular options like Horvath, GrimAge, PhenoAge, and DunedinPACE differ in their ability to forecast factors such as mortality, cognitive decline, and cardiovascular health. These variations can play a key role in tailoring preventive care strategies to individual needs.
Can lifestyle changes really lower my biological age, and how fast?
Yes, making adjustments to your lifestyle can help lower your biological age. Studies reveal that specific actions - like improving your diet, staying active, and adopting healthier habits - can lead to measurable changes in just weeks or months. These shifts work by affecting epigenetic markers, which play a crucial role in how your body ages. While outcomes differ from person to person, sticking to these changes consistently can bring noticeable benefits over time.
How accurate are AI-based biological age tests for predicting disease risk?
AI-powered biological age tests, such as epigenetic clocks, are known for their accuracy, typically showing an average prediction error of around 5.5 years. That said, their reliability isn’t consistent across the board. In some cases, studies have reported discrepancies as large as 25 years. As research advances, these tools are becoming more precise, enhancing their ability to predict disease risks more effectively.
