AI is changing how we understand and manage aging. By analyzing DNA methylation patterns, AI-driven tools can predict biological age with precision, offering insights that go beyond traditional methods like blood tests or general health guidelines. Unlike conventional approaches, which focus on population averages, AI provides personalized strategies tailored to your unique biology.
Key Insights:
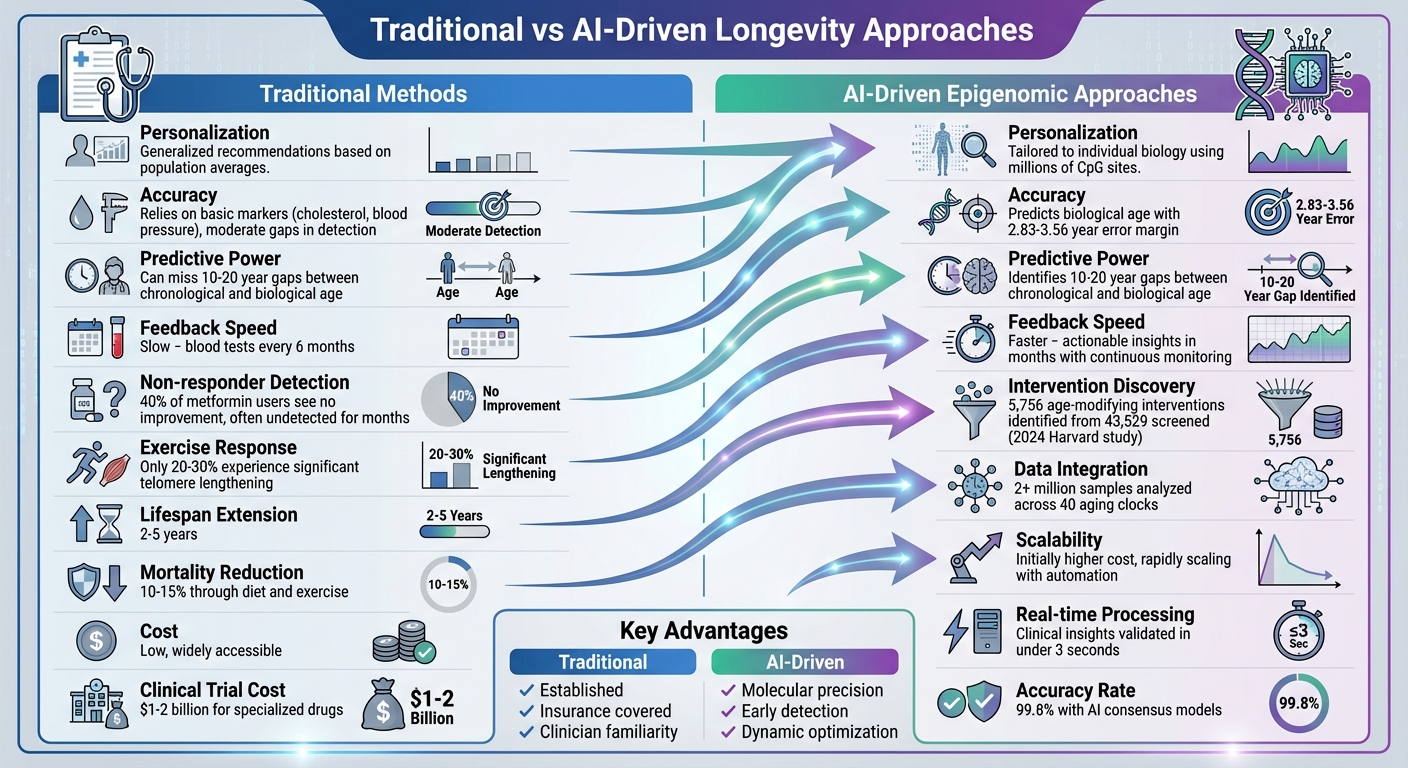
- Traditional Methods: Offer broad health advice (e.g., exercise, diet) but lack personalization and rely on basic markers like cholesterol or blood pressure.
- AI-Driven Epigenomics: Uses massive datasets to create precise "epigenetic clocks", predicting biological age with errors as low as 2.83 years. These tools identify the molecular drivers of aging and suggest specific interventions.
- Real-Time Feedback: AI allows faster adjustments to health plans by analyzing changes in biological age and other markers over months, not years.
- Scalability: AI makes once-costly epigenetic analysis more affordable and accessible, integrating data from wearables, lab results, and more.
Quick Comparison:
| Feature | Traditional Methods | AI-Driven Epigenomics |
|---|---|---|
| Personalization | Generalized recommendations | Tailored to individual biology |
| Accuracy | Relies on basic markers, moderate gaps | Precise biological age predictions |
| Feedback Speed | Slow, months between updates | Faster, actionable insights |
| Cost and Access | Low cost, widely available | Initially higher, but scaling quickly |
AI-powered epigenomics is transforming longevity care, making it possible to not just live longer but live healthier. The future lies in combining traditional methods with these advanced tools for a more complete approach to aging management.
Traditional vs AI-Driven Longevity Approaches Comparison
AI for Longevity: Georg Fuellen on Interventional Data, Synergy Discovery & Generative AI at LSD25
1. Traditional Longevity Approaches
For decades, strategies to promote longer, healthier lives have leaned on population-level guidelines derived from large-scale studies like the Framingham Heart Study, which began in 1948. These studies identified major risk factors - smoking, high blood pressure, high cholesterol, obesity, diabetes, and lack of physical activity - that still serve as the foundation of preventive care today[8]. While these guidelines have been impactful, they fail to account for the unique biological differences among individuals.
Personalization is limited in this model. Most clinicians rely on general demographics and a handful of biomarkers, using tools like the Framingham Risk Score to estimate health risks. But these tools can't capture individual genetic and epigenetic differences. For instance, while physical activity is universally recommended, only 20–30% of people experience significant telomere lengthening - a marker of cellular health - from exercise[4][7]. This one-size-fits-all approach doesn’t account for how differently people respond to the same interventions.
Another drawback is the limited predictive accuracy of traditional methods. These models often rely on chronological age and basic clinical markers like cholesterol levels, ignoring the fact that biological age - the true measure of how "old" your body is - can differ by 10–20 years from your actual age[4][6][7]. This means that even when patients diligently follow guidelines, accelerated cellular aging can still occur if their unique biology isn’t considered[8].
Feedback on interventions is slow and infrequent. Blood tests are typically done every six months, making it hard to adjust treatments quickly. Without real-time monitoring, it’s difficult to identify non-responders. For example, 40% of people taking metformin see no improvement in blood sugar levels, yet this might not be caught until months later[8][5].
While widely accessible, this approach has clear limitations. Public health campaigns based on these guidelines can reach millions at a relatively low cost and have been linked to a 10–15% reduction in all-cause mortality through diet and exercise improvements[4]. However, personalized interventions - like clinical trials for drugs such as rapamycin analogs - are prohibitively expensive, costing $1–2 billion and requiring specialized oversight. As a result, these traditional methods generally extend lifespan by only 2–5 years, far from the potential gains offered by more tailored approaches[2][8].
2. AI-Driven Epigenomic Approaches
AI is reshaping how we measure aging by analyzing DNA methylation patterns that accumulate over time. Unlike older methods, AI integrates massive datasets to provide personalized, real-time insights. These systems pull data from sources like methylation arrays, wearable devices, lab tests, and genetic screenings to create a detailed view of your biological age. For example, platforms like BondMCP – Health Model Context Protocol solve a common issue: fragmented data. Instead of leaving your sleep tracker, lab results, supplement plan, and fitness data isolated in separate apps, BondMCP creates a unified layer that allows all this information to work together. This integration opens the door to deeply personalized aging interventions.
AI takes personalization even further by using deep learning to identify specific aging issues, such as inflammation, mitochondrial dysfunction, or impaired DNA repair. In 2024, researchers at Harvard used ClockBase Agent, an AI system analyzing over 2 million human and mouse samples across 40 aging clocks, to evaluate 43,529 potential interventions. The result? They identified 5,756 interventions with statistically significant age-modifying effects. This level of precision - matching specific treatments to individual aging profiles - is something traditional population-level guidelines simply can't achieve.
With these personalized insights, predictive accuracy also takes a huge leap forward. XAI-AGE, a deep neural network trained on DNA methylation data, predicts biological age with a mean absolute error of just 2.83 years. Compare that to older risk calculators, which might miss a 10–20 year gap between your chronological and biological age[3]. Another advanced aging clock achieved an error margin of 3.56 years, outperforming earlier machine-learning models in both accuracy and correlation with actual health outcomes[3]. For Americans, this means AI could flag accelerated aging far earlier than traditional tests like cholesterol or glucose screenings, allowing for earlier and more effective interventions.
Faster, actionable feedback loops are another game-changer. AI systems can compare baseline and follow-up methylation profiles - usually taken six to twelve months apart - to quickly measure changes in biological age and adjust treatments. When paired with continuous wearable data, these systems can pinpoint how specific interventions affect CpG sites and aging pathways. A BondMCP-based infrastructure automates this process: if your three-month lab results show reduced inflammation and a younger epigenetic age, the AI can instantly update your fitness plan, nutrition strategy, and supplement regimen without you having to juggle multiple platforms.
Finally, scalability becomes a reality. Traditional epigenetic analysis required skilled bioinformaticians to process each sample, which made it slow and expensive. AI changes that by automating tasks like quality control, feature extraction, and risk scoring. This allows thousands - or even millions - of people to receive biological age assessments as the cost of methylation assays continues to drop. Once trained, deep-learning models can generate predictions almost instantly, which is vital for real-time clinical use. BondMCP's Consensus Super AI takes this a step further by validating responses across multiple medically trained models in seconds, delivering clinical-grade insights at scale[1]. This kind of infrastructure makes precision longevity care accessible for employer wellness programs, Medicare initiatives, and consumer health platforms. Instead of static dashboards, users experience dynamic, agent-based optimization tailored to their unique needs.
sbb-itb-f5765c6
Advantages and Disadvantages
Let’s dive into the strengths and weaknesses of traditional methods versus AI-driven epigenomic approaches in longevity care. Traditional tools - like blood panels, imaging, risk scores, and lifestyle guidelines - stand out for their simplicity, familiarity among clinicians, and regulatory acceptance. Tests such as lipid profiles, blood pressure readings, coronary calcium scores, and DEXA scans are well-established for predicting age-related diseases. They’re easy to incorporate into routine clinical workflows, covered by insurance, and backed by decades of epidemiological data. For example, high LDL cholesterol can lead directly to prescribing statins, while elevated blood pressure often results in antihypertensive treatments. However, these methods fall short when it comes to detecting subtle, early signs of biological aging. They also fail to capture the nuanced molecular differences between individuals who may have similar cholesterol or blood pressure levels but vastly different underlying risks [4].
On the flip side, AI-driven epigenomic analysis offers a more advanced lens. By integrating transcriptomics and multi-omics data, it provides a detailed picture of biological age and personalized risk factors that go beyond chronological age and standard lab results [4][3]. These models analyze millions of CpG sites, gene expression patterns, and environmental influences to craft individualized health strategies. Instead of generic advice like "exercise 150 minutes a week", recommendations are tailored to an individual’s unique epigenetic profile, covering areas like nutrition, physical activity, and even pharmacological interventions [4][3]. This shift toward personalization represents a significant leap forward, but it’s not without challenges.
AI-based methods grapple with data quality and standardization issues - epigenomic data is complex and prone to variability. Additionally, deep learning models often lack transparency, functioning as "black boxes." While tools like DeepLIFT in XAI-AGE are making strides in explaining these models, interpretability remains a hurdle [3]. Cost and accessibility are also limiting factors, as comprehensive profiling can be expensive and isn’t widely reimbursed. Ethical and regulatory concerns further complicate the adoption of AI in high-stakes interventions like off-label drug use or gene editing [4][3]. These barriers highlight the need for ongoing advancements to make AI-driven approaches more practical and accessible.
Comparing Traditional and AI-Driven Approaches
Here’s a side-by-side look at how these two approaches stack up across key dimensions:
| Dimension | Traditional Approaches | AI-Driven Epigenomic Approaches |
|---|---|---|
| Personalization | Broad, population-based recommendations (e.g., "exercise 150 minutes/week") with limited tailoring based on age, sex, and a few risk factors [4]. | Highly individualized plans based on molecular and behavioral data, integrating millions of CpG sites and environmental variables [4][3]. |
| Predictive Accuracy | Moderate accuracy for major diseases, limited sensitivity to early biological aging; relies on simple biomarkers and chronological age [4]. | Higher-resolution estimates of biological age and risk using multi-omics and deep learning; stronger links to mortality and morbidity [4][3]. |
| Intervention Feedback | Slow feedback through standard labs (cholesterol, HbA1c, blood pressure) and clinical events; tracks single pathways over months to years [4]. | Fast, precise feedback via aging clocks and pathway analysis; epigenetic changes can reflect systemic aging in months [4][3]. |
| Scalability | Widely available in primary care settings with low costs and minimal infrastructure needs; accessible to most clinicians [4]. | Higher costs and specialized infrastructure required, but scalable once systems are established, with low per-patient computational costs [4][3]. |
Bridging the Gap with Hybrid Models
The most practical path forward may lie in a hybrid approach. Traditional methods can serve as a foundational safety net, while AI-driven tools add value by identifying early biological aging, offering personalized interventions, and enabling dynamic feedback loops. For instance, platforms like BondMCP – Health Model Context Protocol are already addressing this gap. BondMCP integrates fragmented data sources - wearables, lab results, supplements, and fitness metrics - into a unified system. Its Consensus Super AI validates clinical insights across multiple medically trained models in under three seconds, achieving a 99.8% accuracy rate while handling over 2.5 million API calls monthly with 99.9% uptime [1]. This infrastructure enables precision care to scale efficiently across employer wellness programs, Medicare initiatives, and consumer health platforms, making advanced longevity care more accessible than ever.
Conclusion
The transition from traditional approaches in longevity care to AI-powered epigenomic methods marks a major leap in how we evaluate, predict, and manage the aging process. While tools like standard blood panels, imaging, and general lifestyle recommendations remain useful for their simplicity and widespread clinical acceptance, they fall short in delivering the precision and personalization offered by AI-augmented epigenetic clocks. These advanced tools delve into molecular mechanisms - such as DNA methylation patterns and chromatin remodeling - that drive aging at the cellular level, providing insights that go beyond conventional assessments [3]. This shift not only transforms how we measure aging but also lays the groundwork for tailored interventions.
For individuals looking to embrace this new frontier, start with basic clinical evaluations and gradually incorporate AI-driven epigenetic testing. When relevant, polygenic risk scores can add another layer of understanding [3][4]. Work alongside a skilled practitioner to link conventional markers - like elevated inflammatory levels - with epigenomic findings such as accelerated epigenetic aging. From there, focus on foundational lifestyle adjustments: adopt a Mediterranean-style diet, practice moderate caloric restriction, engage in consistent aerobic and resistance exercise, and prioritize quality sleep. These habits have been shown to positively influence epigenetic markers [4][7]. Then, refine these efforts using AI insights tailored to your specific molecular and behavioral profile [3][8].
Recent studies highlight the transformative potential of these advancements. In 2024, Vadim Gladyshev's team at Harvard used the ClockBase Agent AI to analyze over 2 million samples through 40 aging clocks. This effort screened 43,529 interventions and pinpointed 5,756 as likely to modify aging [5]. Such large-scale discoveries, driven by AI's ability to process massive datasets, would have been unthinkable with traditional methods.
Taking personalization further, platforms like BondMCP – Health Model Context Protocol bring together fragmented health data into a unified system for continuous, adaptive care. By integrating information such as epigenetic age, genomic risks, lab results, wearable data, fitness routines, supplements, and sleep patterns, BondMCP enables AI systems to operate cohesively. This means adjustments - for instance, optimizing your training load or refining your supplement plan - can be automatically informed by shifts in your epigenetic age. The result? A dynamic, precise model of your aging process, enabling clinics and digital health platforms to deliver care at a whole new level.
When exploring AI-epigenomic tools, keep these guidelines in mind:
- Look for peer-reviewed evidence supporting the tool's accuracy.
- Choose clinically validated options over consumer-grade alternatives.
- Ensure the tool has been tested in populations similar to you in terms of age, sex, ancestry, and health status.
- Carefully review the privacy policies of the tools you consider [3][4][5].
To stay on track, reassess your epigenetic age and key lab markers every 6–12 months, making adjustments as needed [3][4]. This new era of longevity care is here - personalized, data-driven, and rooted in the molecular realities that shape aging.
FAQs
How does AI enhance the accuracy of predicting biological age compared to traditional methods?
AI is reshaping how we predict biological age by diving deep into epigenomic data - the molecular markers that influence gene activity. Unlike older methods, which often depend on broader, less specific biomarkers, AI pinpoints subtle molecular changes tied to aging.
Using advanced machine learning, these systems interpret complex patterns with remarkable accuracy. The result? Predictions that are not only more precise but also tailored to the individual, providing a sharper understanding of someone's actual biological age.
What challenges come with using AI to analyze epigenomic data for aging and longevity?
AI-powered methods in epigenomic research bring exciting possibilities, but they also come with significant challenges. One of the biggest obstacles is the sheer complexity of epigenomic data. Decoding the intricate biological signals that play a role in aging demands highly advanced tools and methodologies. On top of that, variability in data - stemming from differences among individuals and populations - complicates efforts to develop models that work across the board.
Another pressing issue is the expense of analyzing such data. Effective AI systems require access to large, high-quality datasets, which aren’t always easy or affordable to obtain. Beyond that, ensuring accurate and reproducible results is crucial. Even minor errors in predictions can lead to misleading conclusions, which could derail progress.
That said, advancements in AI and the development of integrative platforms are helping researchers overcome these hurdles, opening doors to more precise and tailored approaches to understanding and enhancing longevity.
How can I start using AI-driven epigenomic tools to improve my longevity strategy?
To start incorporating AI-powered epigenomic tools into your longevity plan, begin by linking your health data sources - like wearables, lab reports, and nutrition apps - into a single, unified platform. Platforms such as BondMCP make this process straightforward by merging your data and delivering personalized health insights.
Once your data is connected, AI can step in to analyze lab results, fine-tune your nutrition, track fitness progress, and evaluate potential health risks in real time. This creates a customized approach to enhancing your healthspan, offering actionable recommendations that align with your long-term wellness goals. The best part? It all operates seamlessly in the background, simplifying decision-making and eliminating the hassle of managing multiple apps or conflicting advice.
