AI-powered digital twins are transforming healthcare by creating virtual models of patients that use real-time data to predict outcomes and personalize treatments. These models integrate information through contextual data fusion from wearables, medical imaging, genomics, and electronic health records to simulate and optimize care strategies. For example, platforms like HeartFlow analyze coronary arteries without invasive procedures, while SOPHiA GENETICS uses oncology-focused digital twins to improve cancer treatment.
Key points:
- Digital twins: Virtual replicas of patients that sync with real-world data.
- AI capabilities: Predict diseases, refine treatments, and simulate outcomes.
- Clinical impact: Reduced hospital readmissions, improved diabetes management, and better surgical planning.
- Challenges: Data integration, validation, and ethical concerns.
AI & Digital Twins: The Next Evolution for Personalised Medicine
sbb-itb-f5765c6
AI Technologies Powering Digital Twins
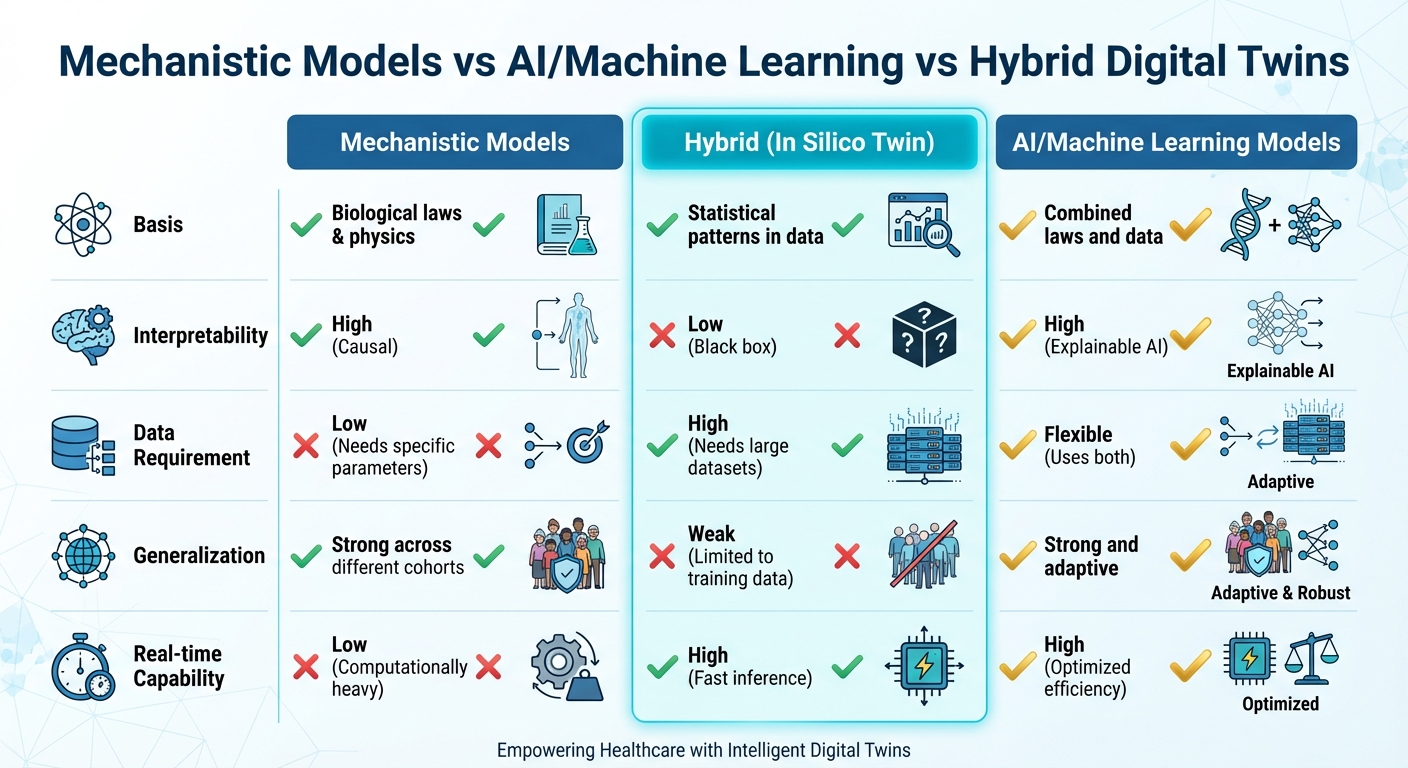
Comparison of Mechanistic Models vs AI Models vs Hybrid Digital Twins in Healthcare
The shift from static patient records to dynamic digital twins relies heavily on advanced AI technologies. These systems sift through vast amounts of health data - ranging from genomic sequences to readings from wearable devices - and transform it into actionable insights that clinicians can use directly in patient care.
Machine Learning and Deep Learning Integration
Machine learning (ML) and deep learning (DL) are at the heart of processing the complex, high-dimensional datasets that digital twins demand. These technologies extract key parameters, such as tumor sizes or genetic risk scores, from diverse data sources, enabling automated and individualized patient modeling [8]. With the integration of cloud-edge architectures, these models achieve real-time processing capabilities at the local level (the edge) while updating global models in the cloud [2].
Reinforcement Learning adds another layer by creating feedback loops that adapt treatment recommendations as a patient’s condition evolves [2]. However, one major drawback of many AI models is their "black-box" nature - it’s often unclear why a model produces certain predictions [4][10]. This limitation has paved the way for hybrid approaches that combine AI with other methods, laying the groundwork for real-time predictive modeling powered by generative AI.
Generative AI for Real-Time Predictive Modeling
Generative AI takes machine learning a step further by enabling real-time simulations of disease progression. This technology enhances digital twins by generating real-time projections of how diseases evolve and how treatments might work. By producing high-quality synthetic data that mirrors real-world disease patterns, generative AI allows clinicians to explore new therapeutic options that haven’t been tested yet [2][7].
For example, SOPHiA GENETICS has introduced a platform for oncology that uses generative AI to simulate disease trajectories. This platform constructs computational replicas of individual patients, enabling real-time modeling of disease progression and survival outcomes [7]. Dr. Pierre Heudel, a Medical Oncologist at Centre Léon Bérard, highlighted the potential of this technology:
Digital twin technology enables integrative visualization of multimodal oncologic data, revealing latent correlations across modalities. By simulating disease trajectories and therapeutic responses within a virtual environment, Digital Twins offer a framework for in silico experimentation, supporting anticipatory and individualized clinical decision making [7].
Generative AI also enhances real-time anomaly detection, identifying subtle changes in a patient’s predicted health state before symptoms become apparent [2]. These capabilities are fueling the growth of the medical digital twin market, which is projected to reach $183 billion by 2031 [11]. This integration of generative AI supports precision care and personalized patient modeling.
Combining Mechanistic Models with AI
While AI excels in predictive accuracy, combining it with mechanistic models addresses the challenge of interpretability. Mechanistic models, grounded in biological laws and physics, provide a clear causal framework, while AI handles the unstructured, high-dimensional data that traditional models struggle to process [2][5]. This hybrid approach merges the strengths of both, offering models that are accurate and interpretable [1][13].
Mechanistic models serve as a "causal core", rooted in well-understood biological principles, while AI accelerates decision-making by substituting complex simulations with real-time predictions [2][5]. Christoph Sadée from the Stanford Center for Biomedical Informatics Research emphasized the importance of this approach:
By combining mechanistic disease modeling with AI, we forecast digital twins that not only predict outcomes but also provide interpretable, patient-specific explanations - a crucial step toward clinical adoption [10].
This blend of AI and mechanistic modeling creates digital twins that are both reliable and easy to understand - qualities essential for their use in clinical settings. The table below highlights how these methods complement each other:
| Feature | Mechanistic Models | AI/Machine Learning Models | Hybrid (In Silico Twin) |
|---|---|---|---|
| Basis | Biological laws & physics | Statistical patterns in data | Combined laws and data |
| Interpretability | High (Causal) | Low (Black box) | High (Explainable AI) |
| Data Requirement | Low (Needs specific parameters) | High (Needs large datasets) | Flexible (Uses both) |
| Generalization | Strong across different cohorts | Weak (Limited to training data) | Strong and adaptive |
| Real-time Capability | Low (Computationally heavy) | High (Fast inference) | High (Optimized efficiency) |
Clinical Applications of AI-Driven Digital Twins
AI-powered digital twins are no longer just theoretical concepts; they're actively reshaping clinical practices. These virtual models of individual patients allow doctors to simulate and test treatments, detect diseases earlier, and refine complex procedures - all without directly impacting the patient. By serving as a "virtual testing ground", digital twins are playing a critical role in improving decision-making in healthcare, starting with therapy planning.
Therapy Planning and Optimization
Digital twins combine biological models with real-time patient data to predict how specific treatments will work for an individual. This enables doctors to experiment with various dosages, drug combinations, and treatment strategies virtually, ensuring the best possible approach before implementation. For example, in diabetes management, these tools have helped improve glucose levels and reduce hypoglycemia through tailored exercise plans.
In cancer care, "cancer avatars" bring together genomic, imaging, and tissue data to forecast a patient's response to treatments like immunotherapy and radiation. This allows doctors to fine-tune treatment plans in advance. A standout example is the DIFAT (digital-heart identification of fat-based ablation targeting) technology, which debuted in 2025. By mapping fat infiltration in the heart, DIFAT reduced ablation volumes for ventricular tachycardia from an average of 7.05 cm³ to just 1.87 cm³, while achieving a 79% match between predicted and actual treatment sites [1].
Reinforcement learning within these systems further refines treatments as a patient's condition changes. This has been particularly effective in oncology. For instance, using digital twins for high-grade gliomas reduced radiation doses by 16.7% without compromising outcomes [1].
Risk Stratification and Early Diagnosis
Digital twins aren't just about treatment - they're also changing how diseases are detected. By analyzing large datasets, such as genomic and proteomic information, these systems can identify early warning signs of disease long before symptoms appear. Wearables and IoT sensors enhance this capability by continuously monitoring subtle physiological changes, sometimes years ahead of clinical symptoms.
Their predictive power has been demonstrated across various conditions, including cancers and neurodegenerative diseases. For instance, digital twins have shown that brain tissue loss in Multiple Sclerosis begins 5–6 years before symptoms develop [1].
In cardiovascular care, the TwinCardio framework, launched in 2025, uses a neural network called TwinNet and IoT sensors for real-time heart health monitoring. This system was developed in response to a 30% rise in heart attacks among adults aged 25–44. By providing accurate risk evaluations and secure data synchronization, TwinCardio has helped reduce cardiac arrest incidents by 60% through predictive alerts [1]. Similarly, the HeartFlow platform has used CT angiography-derived digital twins to estimate coronary flow reserve, cutting down unnecessary invasive procedures and improving care for coronary artery disease patients [3].
Surgical Simulations and Precision Procedures
Digital twins are transforming surgery by providing patient-specific anatomical models based on CT or MRI scans. These models allow surgeons to practice procedures, explore different approaches, and identify potential risks - all in a virtual environment before stepping into the operating room.
In pediatric oncology, the PRIMAGE project used a digital twin framework to achieve near-perfect tumor segmentation accuracy (Dice similarity coefficient of 0.997) while reducing radiologist workload by 93% [1]. This precision enables surgeons to plan with confidence, knowing exactly where to operate and what to avoid.
During surgeries, digital twins also offer real-time monitoring, helping doctors address complications before they escalate. For example, this technology has achieved 89% accuracy in predicting cardiac pro-arrhythmic cardiotoxicity, outperforming traditional animal models, which are only 75% accurate [1]. Additionally, the use of digital twins has been linked to a 25% reduction in hospital readmissions for certain chronic conditions [1].
Challenges and Future Directions
AI-driven digital twins hold immense potential in healthcare, but their path to widespread use is far from straightforward. The intricate nature of human biology, coupled with technical and ethical challenges, means there’s still a long way to go before they become a standard tool in clinical practice.
Data Integration and Interoperability Issues
Creating high-fidelity digital twins requires a seamless flow of data across various biological scales, from molecular processes to organ-level functions. The challenge? These processes operate on vastly different timelines. For example, while gene transcription occurs in minutes, chromatin folding can take days, making real-time synchronization a daunting task [4][12].
Another significant problem is the integration of diverse data sources. Healthcare data comes from everywhere - electronic health records, genomic data, wearable devices, and even social behavior metrics. Each of these uses its own formats and standards, making it tough to unify them into a single, coherent system. As highlighted in a Nature study:
If we want to build the best possible twin of a system we have to replicate the system itself, which does not necessarily advance our understanding. Explanation requires understanding why processes and phenomena happen. [4]
Then there’s the "exposome" gap - the challenge of capturing a person’s entire history of environmental exposures, diet, and social stressors. Many of these factors are rarely measured, leaving critical gaps in the data [4][12]. These shortcomings directly impact the ability to validate digital twin models and ensure their reliability.
Methodology and Validation Barriers
Machine learning models often falter in clinical settings because they’re typically trained on narrow datasets, making them less adaptable to new, real-world scenarios. This lack of adaptability, combined with the opaque nature of many AI models, can erode trust among clinicians who need clear, logical decision-making processes [4][12].
Validation is another sticking point. For digital twins to be clinically viable, they must meet strict validation protocols like ASME V&V 40, which ensures models are accurate enough for medical use [14][1]. A good example is the UVA/Padova Type 1 Diabetes Simulator, which underwent rigorous testing before its artificial pancreas system earned FDA approval [4][14]. However, most digital twin systems haven’t reached this level of scrutiny, especially when dealing with the unpredictable, nonlinear behaviors of biological systems. Even small inaccuracies in data can lead to flawed treatment recommendations, making robust validation a critical hurdle to overcome [4].
Ethics and Privacy Considerations
Ethical and privacy concerns add another layer of complexity. Digital twins rely on continuous access to sensitive personal data, which raises serious questions about how this information is managed and protected. Federated learning offers one potential solution by keeping patient data on local servers and sharing only model updates, but this method is still in its infancy [2].
Bias and fairness are also pressing issues. If AI models are trained on datasets that fail to represent diverse populations, they risk perpetuating existing healthcare inequalities. This makes fairness audits and monitoring for bias essential, though these practices are not yet widespread [2]. Additionally, while digital twins are designed to assist rather than replace clinicians, the line between decision-support tools and autonomous decision-makers can sometimes blur, creating further ethical dilemmas [1][2].
How BondMCP Enables Precision Medicine
BondMCP tackles the challenges of fragmented health data, interoperability issues, and validation barriers by acting as a central intelligence layer. These problems, which often hinder the development of digital twins, stem from disconnected systems that fail to communicate effectively. BondMCP bridges these gaps, creating a cohesive framework that unites the various elements of precision health.
Unified Context for Wearables, Labs, and Interventions
For digital twins to work effectively, they need a complete picture of a patient's health. However, data from wearables, lab tests, and lifestyle interventions often exist in isolated systems. BondMCP solves this by establishing a shared context layer - sometimes called a "digital thread" - that links data from multiple sources, such as multi-omics, electronic health records, IoT devices, and lifestyle metrics. This unified approach allows BondMCP to integrate these diverse data streams into a single, comprehensive view.
With this framework, BondMCP automates the connections between systems. For example, your sleep tracker can inform your fitness coach, your lab results can adjust your supplement plan, and your long-term health goals can guide real-time decisions. By connecting these dots, BondMCP effectively eliminates the fragmentation that often plagues health data systems.
Improving Interoperability and Scalability
One of the major barriers to adopting digital twins is the lack of standardized interoperability frameworks [3]. BondMCP addresses this by providing a structured protocol and SDK, enabling developers to create health-aware AI agents with ease. This setup supports the industry's shift toward agent-based models, where autonomous software agents monitor virtual models and assist clinical teams in real-time [16].
BondMCP's architecture combines edge computing for quick, bedside decisions with cloud resources for large-scale model updates [2]. This dual approach ensures both low-latency responses and scalability, making it possible to deliver precision health solutions on a broader scale. The result is a robust infrastructure capable of supporting dynamic, patient-specific interventions.
Enabling Real-Time Personalized Care
What sets BondMCP apart is its ability to support two-way data flow. This capability allows AI agents to simulate and evaluate multiple treatment options in real-time [16], moving healthcare from reactive treatments to proactive, preventive care. As Tadao Ooka from the University of Yamanashi aptly puts it:
Medical digital twins signify a fundamental transformation in health management, shifting from treating diseases after their occurrence to controlling them before their occurrence [15].
This shift represents a major step forward in personalized medicine, enabling real-time decisions that are tailored to individual patients' needs and circumstances. BondMCP's role in orchestrating this transformation makes it a key player in the evolution of precision health.
Conclusion
AI is reshaping digital twin technology, turning it from static models into dynamic systems that reflect the real-time complexities of human biology [9]. This evolution is steering healthcare toward a proactive approach - spotting risks before symptoms arise and testing AI-powered treatment plans virtually before applying them to real patients. By combining machine learning, generative AI, and mechanistic modeling, researchers are creating "Big AI", which blends the predictive power of AI with mechanistic insights. This combination allows for faster and more reliable patient-specific predictions [6].
The impact of these advancements is already evident. For instance, in a study involving 15,965 patients, an AI-powered ECG monitoring system reduced 90-day all-cause mortality from 4.3% in the control group to 3.6% [15]. At Johns Hopkins University, researchers developed "Geno-DT", a genotype-specific digital twin that uses 3D heart models based on genetic data to improve outcomes for arrhythmogenic right ventricular cardiomyopathy through more effective catheter ablation [17]. These breakthroughs highlight how digital twins are moving from experimental research into real-world clinical use.
Yet, challenges like data fragmentation and interoperability issues remain. Data from wearables, lab tests, and EHR systems often exist in silos, making it difficult to create unified patient models. To overcome this, building a unified health dashboard that integrates multi-omics data, IoT devices, and lifestyle metrics is critical. Such integration would enable the seamless data flow necessary for digital twins to operate effectively.
With 66% of healthcare executives planning to invest in digital twin technologies by 2026 and market growth expected to exceed 30% annually [9], the need for scalable frameworks is clear. These frameworks must prioritize interoperability, real-time data sharing, and scalability to transform fragmented health information into coordinated, patient-focused care. By doing so, digital twins can help shift healthcare toward precision medicine, focusing on optimizing individual health outcomes before illnesses take hold.
FAQs
How accurate are medical digital twins?
The reliability of medical digital twins is still a work in progress. A significant hurdle lies in their dependence on black-box predictive models, which can make understanding and trust in their outcomes more challenging. To address this, hypothesis-driven generative methods, such as multiscale modeling, are seen as key to improving their precision and making them more applicable in clinical settings.
What data do digital twins need from me?
Digital twins use detailed personal health data to create models and simulations of your health. This data includes genetic information, imaging results, physiological metrics, and other important medical details. The more accurate and complete the data you provide, the better the digital twin can offer tailored insights and assist with personalized healthcare.
How is my health data kept private?
Your health data stays secure thanks to advanced security measures, like HTTPS encryption, which protects information during transmission. Cutting-edge technologies, such as blockchain combined with zero-knowledge proofs, add another layer of privacy by verifying data without revealing sensitive details. In the case of digital twin-based precision medicine, strict data protocols are in place to ensure confidentiality and block unauthorized access. This approach allows for personalized healthcare while keeping your information safe.
