Healthcare is shifting towards personalized care, driven by precision medicine and real-time disease simulations. These advancements combine patient-specific data with AI-driven virtual models to predict, prevent, and refine treatments. Here's what you need to know:
- Precision Medicine: Tailors healthcare based on genetics, lifestyle, and medical history, moving beyond generic treatments.
- Real-Time Simulations (Digital Twins): Virtual models use live data from wearables, EHRs, and tests to predict disease progression and optimize treatments.
- Key Technologies: AI, digital twins, and IoT devices enable real-time, patient-specific insights.
- Impact: Reduced procedure times, improved treatment accuracy, and earlier disease detection.
- Challenges: Data integration, bias, computational demands, and regulatory hurdles.
These tools are reshaping healthcare, offering dynamic, data-driven approaches to better manage and prevent diseases.
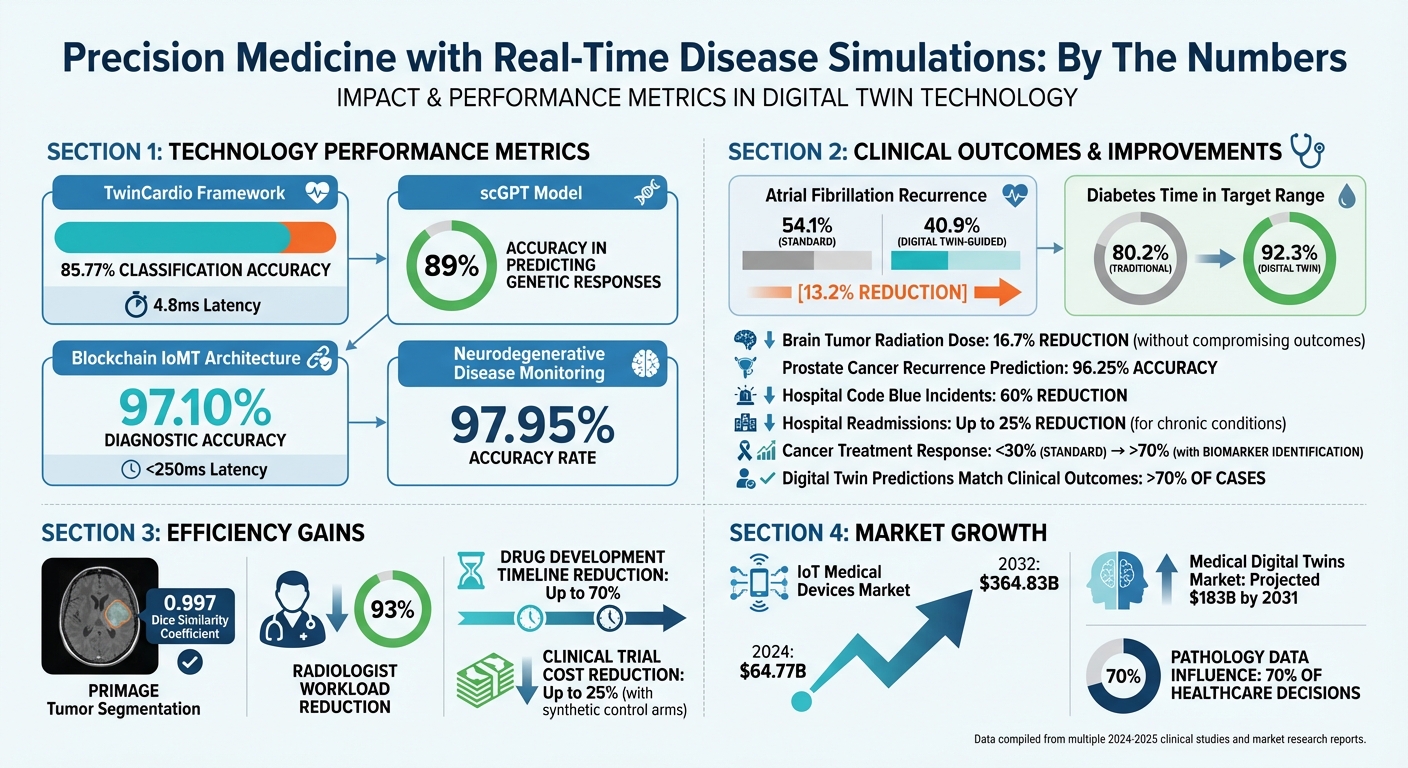
Real-Time Disease Simulations: Key Performance Metrics and Clinical Outcomes in Precision Medicine
How Digital Twins Could End Medical Guesswork
Technologies Behind Real-Time Disease Simulations
Real-time disease simulations bring together three key technologies: AI models that learn from individual health data, digital twins that replicate biological systems, and wearables that provide a steady stream of real-time data. Together, these tools create predictive, personalized simulations that adapt as new information becomes available.
AI and Machine Learning Models
AI models are the engines driving these simulations, processing a wide variety of data like genomics, medical imaging, lab results, and wearable sensor readings. By combining biology-based frameworks with AI's ability to recognize patterns, these models ensure simulations are responsive to data while staying biologically accurate.
Take the TwinCardio framework, introduced in August 2025, as an example. It achieved an impressive 85.77% classification accuracy for real-time cardiac monitoring with a latency of just 4.8 milliseconds [7][4]. This level of responsiveness is crucial for adjusting treatments in real time.
Edge computing plays a pivotal role here, allowing AI models to operate locally - on hospital servers or even wearable devices. This setup enables near-instant responses, which are essential for tasks like detecting arrhythmias or predicting liver function. To handle noisy sensor data, techniques like Bayesian and Kalman filters are employed, making the models more reliable.
Digital Twin Technology
Digital twins act as virtual replicas of an individual's biology, continuously updated with incoming data. These simulations allow for risk-free testing of treatments and interventions, providing a nuanced approach to healthcare.
"An in silico twin (IST) is a continuously learning, mechanistic–AI hybrid model that mirrors an individual's biology and health trajectory, enabling predictive simulation, therapy optimization, and precision clinical decision support."
– Saudi Pharmaceutical Journal [2]
In 2025, the scGPT model demonstrated an innovative way to interpret gene expression profiles, treating genes as "tokens" and their interactions as "sentences." This approach achieved 89% accuracy in predicting genetic responses, surpassing traditional animal models. For cardiac care, therapies guided by digital twins reduced atrial fibrillation recurrence rates to 40.9%, compared to 54.1% with standard treatments [4].
Wearables and IoT Integration
Wearables and IoT devices are the bridge between real-world physiology and virtual simulations. These devices provide continuous data streams, ensuring simulations reflect the user's current health status. In fact, the IoT medical devices market, valued at $64.77 billion in 2024, is expected to grow to $364.83 billion by 2032 [9].
Data processing happens in two stages. First, on-device microcontrollers handle tasks like denoising and feature extraction (e.g., calculating heart rate intervals from ECG data). Then, compressed data is sent to cloud or edge servers to minimize latency.
To improve accuracy, data fusion algorithms combine multiple sources, such as genetic markers, clinical records, and wearable data. These algorithms use weighted fusion and filtering techniques to create a more dynamic and precise model. For instance, in October 2025, a blockchain-enabled IoMT architecture for precision oncology achieved a 97.10% diagnostic accuracy while keeping data processing latency under 250 milliseconds [10]. This kind of real-time monitoring allows digital twins to detect subtle changes in physiology days or even weeks before symptoms appear [4].
Together, these technologies ensure real-time simulations deliver actionable, patient-specific insights seamlessly.
Improving Patient Care with Real-Time Simulations
Real-time simulations are transforming how clinicians approach patient care, enabling treatments that adapt continuously to the patient’s current condition. Instead of relying on static treatment plans, healthcare providers can now make dynamic adjustments using digital replicas of their patients, integrating these updates seamlessly with advanced technologies to deliver highly precise care.
Dynamic Treatment Adjustments
With the help of AI in digital twin-based precision medicine, real-time data now plays a pivotal role in refining treatments on the spot. This closed-loop optimization creates a feedback system between the patient and their digital twin. As new data becomes available, the digital twin updates instantly, allowing clinicians to modify interventions in real time.
In cardiovascular care, for example, a Scenario Engine uses the digital twin to simulate potential outcomes of interventions like adjusting medications or modifying pacing parameters. By testing these changes virtually first, clinicians can minimize the risks associated with trial-and-error methods.
"Virtual interventions such as simulating pacing or drug administration can be tested in the digital environment before being applied to the patient." – Sensors Journal [7]
This method has already shown promising results. For atrial fibrillation, digital twin-guided therapy reduced recurrence rates from 54.1% to 40.9% [4]. In diabetes management, decision support systems using digital twins helped patients maintain their glucose levels within the target range for 92.3% of the time, compared to 80.2% with traditional approaches [4].
Risk Prediction and Prevention
One of the standout benefits of real-time simulations is their ability to detect potential health issues before they become critical. By analyzing data from wearables, lab results, and clinical records, these systems can identify silent conditions like ischemia or arrhythmias weeks in advance, shifting care from reactive to proactive [7].
The results have been game-changing for hospital operations. Digital twin-based Early Warning Systems have cut "code blue" (cardiac arrest) incidents by 60% [4]. In monitoring neurodegenerative diseases, these systems achieved an impressive 97.95% accuracy rate in predicting conditions like Parkinson’s from remote data [4].
This proactive approach also allows clinicians to test high-risk treatments - such as aggressive drug therapies or intense exercise regimens - on a digital twin before applying them to the patient. This ensures safety and effectiveness, paving the way for specialized uses in fields like oncology [4][6].
Case Example: Cancer Treatment Applications
In oncology, Cancer Digital Twins (CDTs) bring together data from multiple sources, including imaging and clinical records, to create virtual models of a patient’s tumor. These models use advanced simulations to predict how various treatments will affect different cancer cell populations [11].
For prostate cancer, machine learning systems have reached a 96.25% accuracy rate in predicting biochemical recurrence [4]. In treating high-grade gliomas (brain tumors), digital twins enabled a 16.7% reduction in radiation dose without compromising clinical outcomes [4].
The PRIMAGE project, focusing on pediatric oncology, demonstrated how digital twins can streamline workflows. It achieved a 0.997 Dice similarity coefficient for tumor segmentation accuracy, while reducing radiologists’ workload by an impressive 93% [4]. By simulating treatments on the digital twin first, clinicians can identify the most effective and safest options.
| Application Area | Key Outcome | Improvement |
|---|---|---|
| Cardiac Arrhythmia | Reduced recurrence rates | 13.2% absolute reduction [4] |
| Diabetes (T1D) | Time in target glucose range | 80.2% → 92.3% [4] |
| Brain Tumor Radiotherapy | Radiation dose optimization | 16.7% dose reduction [4] |
| Hospital Operations | Code blue incidents | 60% reduction [4] |
| Prostate Cancer | Recurrence prediction accuracy | 96.25% [4] |
sbb-itb-f5765c6
How BondMCP Connects Health Data for Real-Time Simulations
Real-time disease simulations rely on one key ingredient: unified data. Without it, even the most sophisticated digital twins can't deliver accurate results. BondMCP steps in as the intelligence layer, bridging fragmented health data and creating a coordinated system that fuels precise, actionable simulations.
Unified Health Data Platform
The problem in precision medicine isn't a lack of data - it’s that the data is scattered across various silos. Think about it: your wearable tracks heart rate, lab results are locked in a clinic portal, genetic data sits in a separate database, and your daily habits aren’t connected to any of them. BondMCP solves this by integrating diverse data sources - from electronic health records (EHRs) and medical imaging to genomic data and real-time wearable sensor streams - all into a unified framework [13][4].
This integration hinges on interoperability standards like FHIR (Fast Healthcare Interoperability Resources) and OMOP (Observational Medical Outcomes Partnership). These standards ensure that data from different systems can work together seamlessly [3]. The result? A cyber-physical system where your physical health and its digital twin stay in sync through the Internet of Medical Things (IoMT). This kind of rapid data exchange is exactly what real-time simulations need, and it enables BondMCP's AI agents to coordinate patient care effortlessly.
Personalized AI Agents for Health Optimization
Once the data is unified, BondMCP's AI agents step in to turn it into actionable insights. These agents don’t just gather information - they coordinate it. They create a closed-loop system that aligns patient routines, interventions, and data [1][7]. For instance, your sleep tracker can inform your fitness coach, your lab results can adjust your supplement plan, and your long-term health goals can guide real-time decisions - all without you juggling multiple apps.
"Digital twins [serve] as cognitive tools that directly support clinical reasoning and decision workflows." – Abigail Silva, 1PerMed Research Group [1]
These AI agents use scenario engines to test "what-if" interventions in a virtual environment before applying them in real life [2][7]. They simplify complex physiological data into clear, guideline-based recommendations for both patients and healthcare providers [7]. For example, when pathology data - responsible for influencing 70% of healthcare decisions - is analyzed using AI agents, treatment response rates can soar from under 30% to over 70% when the right biomarkers are identified [12].
Scalability for Clinics and Developers
BondMCP isn't just designed for individual users - it’s scalable for clinics and developers too. It provides a structured protocol and SDK, eliminating the need to repeatedly build memory systems, agent routing, or toolchains for every new use case. Its edge-to-cloud architecture handles real-time processing on edge devices (as fast as 4.8 milliseconds for ECG signals) while the cloud manages large-scale model retraining and population-level simulations [4][2].
Developers can also take advantage of federated learning, which allows precision health solutions to scale across multiple clinics without transferring sensitive patient data. This ensures privacy while maintaining model accuracy [2]. With a health-focused ontology and plug-and-play orchestration, developers can build once and integrate into an evolving, agent-driven ecosystem - whether they’re a solo innovator or part of a large healthcare network.
The impact of this scalability is undeniable. Digital twin technology has already reduced hospital readmissions by up to 25% for some chronic conditions, while Early Warning Systems using this approach have decreased "code blue" incidents by 60% [4]. BondMCP makes these advancements accessible not just for large research institutions but for any clinic or developer ready to embrace precision healthcare.
Future Developments and Challenges
Precision medicine is advancing rapidly, but making real-time simulations truly patient-specific remains a tough hurdle. These simulations hold a lot of promise, yet scaling them effectively is no small feat. Success hinges on blending hybrid AI frameworks, tackling data bias, and navigating regulatory validation - all while keeping computational demands in check. Let’s dive into how these challenges are being addressed with future-ready solutions.
Hybrid AI Frameworks for Better Accuracy
The future of simulations lies in combining physics-based models with machine learning. Why? Because each has its strengths and weaknesses. Physics-based models excel at capturing established physiological processes but struggle with complex, high-dimensional data. On the other hand, AI models are great at spotting patterns but often lack transparency when it comes to biological plausibility [2][3].
Hybrid frameworks aim to bring the best of both worlds together. By embedding mechanistic constraints into AI models, they ensure predictions align with established biological laws while still making sense of complex, real-time data. A great example is the "tele-stethoscope" prototype developed by researchers at NTT Communication Science Laboratories in 2024. This device combines ECG data with heart sounds and vibrations, using cardio-specific rules to create internal heart images from surface sounds. This approach has significantly improved diagnostic accuracy [6].
"Generative models have the potential to simulate complex biological functions and how the body will respond to different scenarios" – Kunio Kashino, AI Specialist at NTT Communication Science Laboratories [6]
These hybrid models also incorporate advanced techniques like Bayesian estimation and Kalman filtering to handle uncertainty. This allows them to adapt to incoming sensor data, even when faced with noise or gaps in the information [7].
Addressing Computational and Data Bias Issues
One major challenge is data bias. When training datasets are not diverse enough, models can perform inconsistently across different demographic groups. To counter this, developers are using fairness metrics like equalized odds and demographic parity, along with continuous drift monitoring to catch performance issues early [2].
Federated learning is another solution. By enabling institutions to share insights without directly sharing data, this approach enhances privacy and broadens the diversity of training datasets [2][7]. For rare diseases, where data is often limited, meta-learning helps by identifying patterns between well-studied and less-understood conditions, improving the accuracy of predictions [6].
Then there’s the issue of computational demands. High-fidelity heart models, for instance, require sub-millisecond response times to be useful in clinical settings. To meet this requirement, developers are turning to reduced-order surrogate methods, like Neural ODEs, and edge-to-cloud computing. These systems handle low-latency tasks at the bedside while reserving large-scale model retraining for the cloud [2][4][7].
Building trust is also key. Explainable AI (XAI) tools like SHAP and LIME are being integrated into these models. These tools make it easier to visualize a model’s decision-making process, ensuring predictions align with known biology and earning the trust of clinicians [2].
Scaling Real-Time Simulations in Clinical Settings
Even with advancements in AI and data handling, integrating real-time simulations into clinical workflows comes with its own set of challenges. Regulatory validation is a significant barrier, as these simulations are often classified as medical devices. This means they must undergo extensive validation before they can be used in hospitals. Programs like the FDA’s Medical Device Development Tools (MDDT) are helping to standardize this process, but progress is slow [3][5].
Digital twins offer a promising way forward, especially in clinical trials. By serving as synthetic control arms, they could cut trial costs by as much as 25% [5].
Data interoperability is another hurdle. Fragmented data from electronic health records (EHRs), wearables and imaging systems often slows down adoption [3][2]. However, projects like PRIMAGE are proving that these challenges can be overcome. As of November 2025, PRIMAGE successfully integrated imaging biomarkers and AI to achieve a tumor segmentation accuracy of 0.997 (Dice similarity coefficient). This reduced radiologists' manual workload by 93% while maintaining high precision in neuroblastoma and DIPG cases [4]. Results like these show the potential for scaling, provided infrastructure, regulatory processes, and clinical workflows are aligned.
| Challenge | Strategy for Mitigation | Expected Outcome |
|---|---|---|
| Computational Cost | Reduced-order models & Edge computing | Real-time simulation with low latency [4][7] |
| Data Bias | Federated Learning & Fairness metrics | Equitable outcomes across diverse populations [2] |
| Data Scarcity | Meta-learning & Generative AI | Better modeling for rare diseases [6] |
| Model Opacity | Explainable AI (XAI) & Hybrid models | Greater clinician trust and transparency [2][3] |
Conclusion
Real-time disease simulations are transforming healthcare by shifting the focus from reactive treatments to proactive, patient-centered care. By leveraging digital twins and hybrid AI systems, these simulations can predict disease progression weeks in advance, allowing for earlier interventions. This represents a major evolution: instead of simply managing diseases after they appear, healthcare providers can now work to prevent complications using continuous physiological modeling.
The numbers speak for themselves. Retrospective studies on cardiac resynchronization therapy show that digital twin predictions matched actual clinical outcomes in over 70% of cases [1]. In drug development, AI-powered simulations are expected to cut development timelines by up to 70% [14]. Meanwhile, the market for medical digital twins is projected to grow to $183 billion by 2031 [8]. These advancements aren’t theoretical - they’re already being applied in clinics and research labs around the world.
Experts emphasize the transformative potential of this technology:
"Digital twins are virtual representations of health and disease processes that can integrate real-time data and simulations to predict, prevent, and personalize treatments." - Genome Medicine [8]
Still, for these simulations to reach their full potential, seamless data integration is critical. Currently, much of a patient’s health data remains fragmented and siloed. Platforms like BondMCP are stepping in to solve this problem by unifying disparate data sources, enabling real-time, coordinated care. These tools allow healthcare providers to make informed decisions without overhauling existing systems, aligning perfectly with the vision of precision medicine.
Looking ahead, the future of healthcare will depend on a fully connected ecosystem where health data flows freely, supporting both wellness and disease prevention. Real-time simulations provide the predictive insights, while integrated platforms ensure those insights translate into actionable, effective care.
FAQs
How do real-time disease simulations enhance patient care?
Real-time disease simulations are reshaping how patient care is delivered by leveraging dynamic, personalized data - like details from wearable devices or electronic medical records. These simulations model how diseases progress and predict future health outcomes, giving healthcare providers an edge in spotting early warning signs and customizing treatments. The ability to adjust care plans on the fly means interventions can be more timely and effective.
Another key benefit is how these simulations enhance treatment personalization. By forecasting how an individual might respond to various therapies, they reduce the guesswork that often comes with trial-and-error methods. This not only minimizes side effects but also allows for safer, more targeted care. Real-time disease modeling empowers healthcare professionals to make proactive, data-driven decisions, setting a higher bar for precision medicine.
How do wearables and IoT devices contribute to precision medicine?
Wearables and IoT devices play an important role in pushing precision medicine forward. These gadgets collect real-time data on vital signs, activity levels, sleep patterns, and other health metrics around the clock. The result? A constantly updated, personalized health profile that healthcare providers and AI systems can use to simulate how diseases might progress and even predict potential health challenges.
When this data is fed into digital models, it transforms healthcare from a one-size-fits-all approach to a more proactive, personalized strategy. Treatments and interventions can be adjusted in real time, fine-tuning care to address each individual’s specific needs and improving overall outcomes.
What are the main challenges in using digital twins for healthcare?
Creating and using digital twins in healthcare isn't without its hurdles. One major challenge lies in accurately simulating human physiology and diseases. This requires pulling together a massive amount of diverse, high-quality data. The complexity of this task makes it tough to build reliable, real-time models tailored to individual patients.
Another obstacle is interoperability - getting different devices, platforms, and data sources to work together seamlessly. Health data is often scattered across systems, which can disrupt the smooth communication needed for effective simulations. On top of that, there are strict regulatory and validation requirements to tackle. Digital twins must meet high standards for safety, accuracy, and privacy before they can be trusted in clinical environments. The constantly changing regulatory landscape and the need to update models as a patient’s health evolves add even more layers of complexity.
Addressing these challenges will require better computational power, improved data-sharing protocols, and more refined regulatory frameworks. These advancements are key to tapping into the full potential of digital twins for personalized medicine.
